Abstract
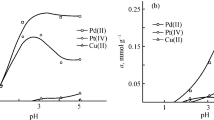
The possibility of selective recovery of palladium(II) and platinum(IV) in the batch mode with heterochain complexing sorbents of polycondensation type from hydrochloric acid solutions simulating spent leaching solutions of spent industrial catalysts and mother liquor after ammonium hexachloroplatinate precipitation was examined. At 19°C and phase contact time of 2 h, the sorbent with 1-(2-aminoethyl)piperazine and dimethylene sulfide functional groups recovers more than 99.9% of palladium(II) of the simulated solution from leaching APK-2 catalyst (sorption capacity ЕPd = 501 mg g–1) and 97–98% of platinum(IV) from the simulated solution of leaching the platinum–nickel catalyst (EPt = 86 mg g–1) with the selective separation of Pd(II) from Al(III) and of Pt(IV) from Al(III) and Ni(II). Polymethylene monosulfide at 80°C and phase contact time of 3 h recovers almost completely recovers palladium(II) (ЕPd = 34 mg g–1) and 95% of platinum(IV) (EPt = 6 mg g–1) from the simulated mother liquor with selective separation of the platinum metals from the concomitant elements Cu(II), Zn(II), Fe(III), and Sn(IV).


Similar content being viewed by others
REFERENCES
Cowley, A., PGM Market Report, February 2020, Johnson Matthey PLC, 2020.
Informatsionno-tekhnicheskii spravochnik po nailuchshim dostupnym tekhnologiyam ITS 14-2016. Proizvodstvo dragotsennykh metallov (ITS 14-2016 Information and Technical Handbook on Best Available Technologies. Production of Precious Metals), Moscow: Byuro NDT, 2016, pp. 3–8, 29.
Il’yashevich, V.D., Pavlova, E.I., Zherebtsova, O.V., Sirotina, D.Yu., Lukina, K.V., and Krivosheev, N.O., Abstracts of Papers, XXII Mezhdunarodnaya Chernyaevskaya konferentsiya po khimii, analitike i tekhnologii platinovykh metallov (XXII Int. Chernyaev Conf. on Chemistry, Analytical Chemistry, and Technology of Platinum Metals), Moscow, 2019, pp. 85.
Blokhin, A.A., Abovskii, N.D., Murashkin, Yu.V., and Mikhailenko, M.A., in Tsvetnye metally Sibiri-2009: Sbornik dokladov Pervogo mezhdunarodnogo kongressa (Nonferrous Metals of Siberia 2009: Proc. First Int. Congr.), Krasnoyarsk: Verso, 2009.
Yahorava, V. and Kotze, M., J. S. Afr. Inst. Min. Metall., 2014, vol. 114, no. 2, pp. 173–181.
Nikoloski, A.N. and Ang, K.-L., Miner. Process. Extr. Metall. Rev., 2014, vol. 35, no. 6, pp. 369–389. https://doi.org/10.1080/08827508.2013.764875
Informatsionno-tekhnicheskii spravochnik po nailuchshim dostupnym tekhnologiyam ITS 14-2016. Proizvodstvo dragotsennykh metallov (ITS 14-2016 Information and Technical Handbook on Best Available Technologies. Production of Precious Metals), Moscow: Byuro NDT, 2016, pp. 19–21, 49.
Ehrlich, H.V., Buslaeva, T.M., and Maryutina, T.A., Russ. J. Inorg. Chem., 2017, vol. 62, no. 14, pp. 1797–1818. https://doi.org/10.1134/S0036023617140030
Tatarnikov, A.V., Sokolskaya, I., Shneerson, Ya.M., Lapin, A.Yu., and Goncharov, P.M., Platinum Met. Rev., 2004, vol. 48, no. 3, pp. 125–132. https://doi.org/10.1595/147106704X1667
Bogdanov, V.I., Baraboshkin, V.E., and Grokhovskii, S.V., Drag. Met. Drag. Kamni, 2005, no. 8, pp. 86–113.
Dal’nova, O.A., Baranovskaya, V.B., Dal’nova, Yu.S., and Karpov, Yu.A., J. Anal. Chem., 2018, vol. 73, no. 3, pp. 221–227. https://doi.org/10.1134/S1061934818030036
Zolotov, Yu.A., Tsizin, G.I., Dmitrienko, S.G., and Morosanova, E.I., Sorbtsionnoe kontsentrirovanie mikroelementov iz rastvorov: primenenie v neorganicheskom analize (Sorption Preconcentration of Microelements from Solutions: Use in Inorganic Analysis), Moscow: Nauka, 2007.
Patent RU 2205237, Publ. 2003.
Semenov, D.G., Afonin, V.M., de Vekki, D.A., and Simanova, S.A., Izv. Sankt-Peterb. Gos. Tekhnol. Inst. (Tekh. Univ.), 2019, vol. 56, no. 50, pp. 3–15.
Rafikov, S.R., Aleev, R.S., Masagutov, R.M., Danilov, V.T., and Dal’nova, Yu.S., Russ. Chem. Bull., 1982, vol. 31, pp. 1452–1453. https://doi.org/10.1007/BF00954173
Saldadze, K.I. and Kopylova-Valova, V.D., Kompleksoobrazuyushchie ionity (kompleksity) (Complexing Ion-Exchange Resins), Moscow: Khimiya, 1980.
Anpilogova, G.R., Cand. Sci. (Chem.) Dissertation, Ufa, 1997.
Ginzburg, S.I., Ezerskaya, N.A., Prokof’eva, I.V., Fedorenko, N.V., Shlenskaya, V.I., and Bel’skii, N.K., Analilticheskaya khimiya platinovykh metallov (Analytical Chemistry of Platinum Metals), Moscow: Nauka, 1972.
Blokhin, A.A., Abovskii, N.D., and Murashkin, Yu.V., Russ. J. Appl. Chem., 2007, vol. 80, no. 7, pp. 1058–1062. https://doi.org/10.1134/S1070427207070087
Anpilogova, G.R., Khisamutdinov, R.A., Golubyatnikova, L.G., and Murinov, Yu.I., Russ. J. Appl. Chem., 2016, vol. 89, no. 2, pp. 206–211. https://doi.org/10.1134/S1070427216020075
Kalinina, L.S., Motorina, M.A., Nikitina, N.I., and Khachapuridze, N.A., Analiz kondensatsionnykh polimerov (Analysis of Condensation Polymers), Moscow: Khimiya, 1984.
Markov, V.K., Vernyi, E.A., Vinogradov, A.V., Elinson, S.V., Klygin, A.E., and Moiseev, I.V., Uran. Metody ego opredeleniya (Uranium. Determination Methods), Moscow: Atomizdat, 1964.
Tikhonov, V.N., Analiticheskaya khimiya alyuminiya (Analytical Chemistry of Aluminum), Moscow: Nauka, 1971, pp. 67–69.
Myshlyaeva, L.V. and Maksimova, T.G., Zh. Anal. Khim., 1968, vol. 23, no. 10, pp. 1584–1586.
Vasil’ev, V.P., Analiticheskaya khimiya (Analytical Chemistry), Gravimetricheskii i titrimetricheskii metody analiza (Gravimetric and Titrimetric Methods of Analysis), Moscow: Vysshaya Shkola, 1989.
Anpilogova, G.R., Aleev, R.S., Afzaletdinova, N.G., Khisamutdinov, R.A., Shishlov, N.S., and Murinov, Yu.I., Zh. Neorg. Khim., 1995, vol. 40, no. 3, pp. 466–471.
Shestakov, V.A., Malofeeva, G.I., Petrukhin, O.M., Shiryaeva, O.A., Kolonina, L.N., Marcheva, E.V., Murinov, Y.I., Bikbaeva, G.G., Nikitin, Y.E., and Zolotov, Y.A., J. Anal. Chem. USSR, 1981, vol. 36, no. 9, pp. 1250–1256.
Petrukhin, O.M., Malofeeva, G.I., Nefedov, V.I., Salyn, Y.V., Marcheva, E.V., Shestakov, V.A., Shiryaeva, O.A., Murinov, Y.I., Nikitin, Y.E., and Zolotov, Y.A., J. Anal. Chem. USSR, 1983, vol. 38, no. 2, pp. 196–201.
ACKNOWLEDGMENTS
The authors are grateful to the Center of Shared Use “Chemistry,” Ufa Institute of Chemistry, Ufa Federal Research Center, Russian Academy of Sciences and to the Regional Center of Shared Use “Agidel,” Ufa Federal Research Center, Russian Academy of Sciences for performing elemental analysis of the sorbents.
Funding
The study was performed within the framework of the government assignment (theme no. АААА-А20-120012090027-6).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
Additional information
Translated from Zhurnal Prikladnoi Khimii, No. 3, pp. 324–331, January, 2021 https://doi.org/10.31857/S0044461821030063
Rights and permissions
About this article
Cite this article
Anpilogova, G.R., Murinov, Y.I. Recovery of Palladium(II) and Platinum(IV) with Heterochain Complexing Sorbents from Solutions Simulating Leaching Solutions of Spent Industrial Catalysts and Spent Refining Solution. Russ J Appl Chem 94, 310–316 (2021). https://doi.org/10.1134/S107042722103006X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107042722103006X