Abstract
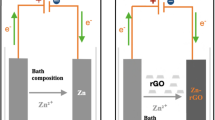
In this study, the corrosion behavior of Zircaloy was investigated in the presence and absence of copper–graphene nanocomposites coating. The coating was prepared employing Hummers’ and electrochemical reduction methods. The morphology of copper–graphene nanocomposites coating was studied using scanning electron microscopy (SEM). Corrosion behavior was investigated employing dynamic polarization and electrochemical impedance spectroscopy (EIS) tests in a solution containing lithium hydroxide (LiOH), boric acid (H3BO3), and deionized water. The results showed that corrosion resistance of Zircaloy increased with introduction of copper–graphene nanocomposites coatings. The lowest corrosion rate was attained in the Zircaloy with copper–graphene nanocomposites coating (corrosion rate: 0.040 mm/year). An approximately 20 times decrease in the corrosion rate was observed in the Zircaloy with copper–graphene nanocomposites coating when compared to the un-coated Zircaloy (corrosion rate: 0.831 mm/year).
Similar content being viewed by others
References
Nikulin, S.A., Rozhnov, A.B., Koteneva, M.V., Belov, V.A., and Komissarov, A.A., Russ. Metallurgy (Metally) 2012, vol. 10, pp. 906–910.
Filippov, V.P., Bateev, A.B., Lauer, Y.A., Dykhuis, A.F., and Short, M.P., Hyperfine Interactions 2018, vol. 239, no. 21, pp. 1–8.
Zohuri, B., In Small Modular Reactors as Renewable Energy Sources, Springer, Cham, 2019.
Waterside Corrosion of Zirconium Alloys in Nuclear Power Plants, IAEA-TECDOC-996, International Atomic Energy Agency, Vienna, Austria, 1998.
Qin, L., Bian, D., Zhao, Y., Xu, X., and Guo, Y., Russ. J. Appl. Chem., 2017, vol. 90, pp. 811–817.
Nezamdoust, S. and Seifzadeh, D., Progress in Organic Coatings 2017, vol. 109, pp. 97–109.
Tseluikin, V.N. and Koreshkova, A.A., Russ. J. Appl. Chem. 2016, vol. 89, pp. 1027–1030.
Mohammadi, S. and Roohi, H., Corrosion Engineering, Science and Technology 2018, vol. 53, pp. 422–433.
Karanveer, S. Aneja, MallikaBöhm, H.L., and Khanna, A.S., Flat. Chem. 2017, vol. 1, pp. 11–19.
Lianbin Zhang, Guoying Chen, Mohamed Nejib Hedhili, Hongnan Zhang, and Peng Wang, Nanoscale 2012, vol. 4, pp. 7038–7045.
Xie, G., Forslund, M., and Pan, J., Direct Electrochemical Synthesis of Reduced Graphene Oxide (RGO)/Copper Composite Films and Their Electrical/Electroactive Properties, ACS Appl. Mater 2014.
ASTM-B-614–05, Standard Practice for Descaling and Cleaning Zirconium and Zirconium Alloy Surfaces.
Ai, J., Chen, Yingzi, and Urquidi-Macdonald, Mirna, and Macdonald, Digby, J. Nuclear Materials 2008, vol. 379, pp. 162–168.
Li, Q., Yang, X., Zhang, L., and Wang, J., Alloy Compd. J. 2009, vol. 482, pp. 339–344.
Cesiulis, H., Tsyntsaru, N., Ramanavicius, A., and Ragoisha, G., The Study of Thin Films by Electrochemical Impedance Spectroscopy, Springer International Publishing, Switzerland. doi 10.1007/978-3-319-30198-3-1, 2016.
Vrsalović, L., Gudić, S., Indian J. Chem. Techn. 2012, vol. 19, pp. 96–102.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Adelkhani, H., Tayebi, M. Improve Corrosion Resistance of Zircaloy by Copper–Graphene Nanocomposite Coatings. Russ J Appl Chem 91, 2052–2057 (2018). https://doi.org/10.1134/S1070427218120182
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427218120182