Abstract
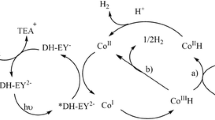
Thiolate complexes Co(bpy)(pyS)2 (M) were synthesized, the effect of reaction conditions (such as pH values, solvents, electron donor, photosensitizers, catalyst, and concentrations) on catalytic performance of catalyst M was studied and the optimum reaction conditions were selected. The results of catalytic performance imply that complexes M showed good catalytic activity when concentration of catalyst M was 2.5 μM, concentration of fluorescein was 2.0 mM, pH value was 11.6, electron donor was triethylamine (5%), and the solvent was EtOH/H2O (1: 1 V/V). The catalysis system presented the best performance and hydrogen production reached 369.2 μmol h–1 after reacting for 15 h; the life of catalyst M was 41 h investigated under the optimum conditions.
Similar content being viewed by others
References
Graetzel, M., Acc. Chem. Res., 1981, vol. 14, pp. 376–384.
Esswein, A.J. and Nocera, D.G., Chem. Rev., 2007, vol. 107, pp. 4022–4047.
Wang, M., Na, Y., Gorlov, M., Sun, L., et al., Dalton Trans., 2009, pp. 6458–6467.
Sakai, K. and Ozawa, H., Coord. Chem. Rev., 2007, vol. 251, pp. 2753–2766.
Cline, E.D., Adamson, S.E., and Bernhard, S., Inorg. Chem., 2008, vol. 47, pp. 10378–10388.
Elvington, M., Brown, J., Arachchige, S.M., et al., J. Am. Chem. Soc., 2007, vol. 129, pp. 10644–10645.
Ozawa, H., Haga, M.-a., and Sakai. K., J. Am. Chem. Soc., 2006, vol. 128, pp. 4926–4927.
Lei, P., Hedlund, M., Lomoth, R., et al., J. Am. Chem. Soc., 2007, vol. 130, pp. 26–27.
Razavet, M., Artero, V., and Fontecave, M., Inorg. Chem., 2005, vol. 44, pp. 4786–4795.
Hu, X.L., Cossairt, B.M., and Brunschwig, B.S., et al., Chem. Commun., 2005, vol. 37, pp. 4723–4725.
Connolly, P. and Espenson J.H., Inorg. Chem., 1986, vol. 25, pp. 2684–2688.
Dempsey, J.L., Brunschwig, B.S., and Winkler, J.R., et al., Acc. Chem. Res., 2009, vol. 42, pp. 1995–2004.
Dempsey, J.L., Winkler, J.R., and Gray, H.B., J. Am. Chem. Soc., 2010, vol. 132, pp. 1060–1065.
Lazarides, T., McCormick, T., Eisenberg, R., et al., J. Am. Chem. Soc., 2009, vol. 131, pp. 9192–9214.
McCormick, T.M., Calitree, B.D., Orchard, A., et al., J. Am. Chem. Soc., 2010, vol. 132, pp. 15480–15483.
Probst, B., Kolano, C., Hamm, P., et al., Inorg. Chem., 2009, vol. 48, pp. 1836–1843.
Probst, B., Rodenberg, A., Guttentag, M., et al., Inorg. Chem., 2010, vol. 49, pp. 6453–6460.
Paul, T., Miller, P.L., and Strathmann, T.J., Env. Sci. & Techn., 2007, vol. 41, no. 13, pp. 4720–4727.
McCormick, T.M., Calitree, B.D., Orchard, A., et al., J. Amer. Chem. Soc., 2010, vol. 132, no. 44, pp. 15480–15483.
Curtin, P.N., Tinker, L.L., Burgess C M, et al., Inorg. Chem., 2009, vol. 48, no. 22, pp. 10498–10506.
Yorozu T, Hoshino M, Imamura M, et al., J. Phys. Chem., 1982, vol. 86, no. 22, pp. 4422–4426.
Elvington M, Brewer K J., Inorg. Chem., 2006, vol. 45, no. 14, pp. 5242–5244.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Cai, X., Li, G., Yang, Y. et al. Cobalt thiolate complexes catalyst in noble-metal-free system for photocatalytic hydrogen production. Russ J Appl Chem 89, 1506–1511 (2016). https://doi.org/10.1134/S1070427216090196
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427216090196