Abstract
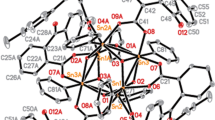
Chemists are pretty much acquainted with coordination compounds bearing carboxylate moieties. Nevertheless comprehensive literature on the subject correlating activity and toxicity with varied organotin(IV) carboxylates containing a diverse array of ligand moieties under identical experimental parameters is lacking. In view of the exciting structural and biological importance attributed to organotin(IV) compounds, the current research work involved synthesizing di- and triorganotin(IV) analogues of carboxylate ligand with hexyl aliphatic, aromatic and alicyclic backbone. The new organotin(IV) complexes with cyclohexane carboxylic acid (L1), benzoic acid (L2), and heptanoic acid (L3) were synthesized by the reaction of di- and triorganotin(IV) salts in the presence of trimethylamine as base. The coordination mode of ligand, structural confirmation and geometry assignment of complexes were established using FT-IR, 1H NMR and 13C NMR spectra. The IR data implied that in both di- and triorganotin(IV) carboxylates, the ligand moiety—(COO) acted as a bidentate group in solid state. 1H and 13C NMR data revealed that tri and diorganotin(IV) complexes occurred as monomeric entities with tetrahedral geometry around tin in solution. The synthesized complexes were also evaluated for their antibacterial and antifungal activity. The results showed that synthesized complexes have greater inhibition potential against different strains of bacteria and fungi compared with the corresponding free ligands. Triorganotin(IV) compounds were found to be more potent against tested bacterial and fungal strains than that of diorganotin(IV) derivatives, having moderate antioxidant activity. Most of the organotin(IV) complexes of benzoic acid were found to be more potent against strains of bacteria and fungi than that of corresponding complexes of other ligands showing that for a biological activity to be shown by a complex, the role of ligand was crucial.



Similar content being viewed by others
REFERENCES
Hussain, S., Ali, S., Shahzadi, S., Sharma, K.S., Qanungo, K., and Shahid, K., Bioinorg. Chem. Appl., 2014, vol. 2014, p. 1. https://doi.org/10.1155/2014/959203
Matela, G., and Aman, R., Cent. Eur. J. Chem., 2012, vol. 10, p. 1. https://doi.org/10.2478/s11532-011-0107-6
Rehman, Z., Barsan, M.M., Wharf, I., Muhammad, N., Ali, S., Meetsma, A., and Butler, I.S., Inorganica Chim. Acta., 2008, vol. 361, p. 3322. https://doi.org/10.1016/j.ica.2008.05.006
Chilwal, A., Malhotra, P., and Narula, K., J. Therm. Anal. Calorim., 2013, vol. 114, p. 345. https://doi.org/10.1007/s10973-012-2880-7
Kumar, M., Abbas, Z., Tuli, H. S., and Rani, A., Curr. Pharmacol. Rep., 2020, vol. 6, p. 167. https://doi.org/10.1007/s40495-020-00222-9
Rehman, A., Hussain, M., Rehman, Z., Ali, S., Rauf, A., Nasim, F.H., and Helliwell, M., Inorganica Chim. Acta, 2011, vol. 370, p. 27. https://doi.org/10.1016/j.ica.2011.01.007
Mazhar, M., Khan, M.K., Mahon, M.F., and Molloy, K.C., J. Organomet. Chem., 2004, vol. 689, p. 899. https://doi.org/10.1016/j.jorganchem.2003.12.019
Shaheen, F., Ali, S., Rosario, S., and Shah, N.A., J. Coord. Chem., 2014, vol. 67, p. 1851. https://doi.org/10.1080/00958972.2014.908187
Attanzio, A., D’Agostino, S., Busà, R., Frazzitta, A., Rubino, S., Girasolo, M. A., Sabatino, P., and Tesoriere, L., Molecules, 2020, vol. 25, p. 859. https://doi.org/10.3390/molecules25040859
Gielen, M., Biesemans, M., and Willem, R., Appl. Organomet. Chem., 2005, vol. 19, p. 440. https://doi.org/10.1002/aoc.771
Hosseiny, M.S., Rounaghi, G.H., and Sadeghian, H., Russ. J. Inorg. Chem., 2011, vol. 56, p. 816. https://doi.org/10.1134/S0036023611050093
Rehman, A., Choudhary, M. I., Thomsen, W.J., Bioassay Techniques for Development, Amsterdam: Harwood Academic Publishers, 2001, p. 9
Clinical and Laboratory Standards Institute. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically: Approved Standard, 2015, 10th ed Wayne, PA: Clinical & Laboratory Standards Institute; CLSI document M7-A10.
Shahzadi, S., Shahid, K., and Ali, S., Russ. J. Coord. Chem., 2007, vol. 33, p. 403. https://doi.org/10.1134/S1070328407060048
Yin, H.D. and Sheng, C.X., Appl. Organomet. Chem., 2006, vol. 20, p. 283. https://doi.org/10.1002/aoc.1048
Shah, F.A., Shahzadi, S., Ali, S., Molloy, K.C., and Ahmad, S., Russ. J. Coord. Chem., 2009, vol. 35, p. 896. https://doi.org/10.1134/S1070328409120069
Rehman, W., Baloch, M.K., Badshah, A., and Ali, S., Russ. J. Coord. Chem., 2005, vol. 31, p. 856. https://doi.org/10.1007/s11173-005-0181-4
Matela, G. and Aman, R., Cent. Eur. J. Chem., 2012, vol. 10, p. 1. https://doi.org/10.2478/s11532-011-0107-6
Sirajuddin, M., Ali, S., McKee, V., Zaib, S., and Iqbal, J., RSC Adv., 2014, vol. 4, p. 57505. https://doi.org/10.1039/C4RA10487K
Waseem, D., Butt, A.F., Haq, I., Bhatti, M.H., and Khan, G.M., DARU J. Pharm. Sci., 2017, vol. 25, p. 1. https://doi.org/10.1186/s40199-017-0174-0
Lockhart, T.P., Manders, W.F., and Holt, E.M., J. Am. Chem. Soc., 1986, vol. 108, p. 6611. https://doi.org/10.1021/ja00281a026
Lockhart, T.P. and Manders, W.F., Inorg. Chem., 1986, vol. 25, p. 892. https://doi.org/10.1021/ic00227a002
Sharma, S., Meena, R., Singh, R.V., and Fahmi, N., Main Group Met. Chem., 2016, vol. 39, p. 31. https://doi.org/10.1515/mgmc-2015-0030
Hussain, S., Ali, S., Shahzadi, S., Tahir, M.N., and Shahid, M., J. Coord. Chem., 2015, vol. 68, p. 2369. https://doi.org/10.1080/00958972.2015.1046849
Graisa, A.M., Husain, A.A., Al-Mashhadani, M.H., Ahmed, D.S., Adil, H., and Yousif, E., J. Serambi Eng., 2022, vol. 7, p. 2631. https://doi.org/10.32672/jse.v7i1.3825
Liu, K., Chang, G., Yan, H., Li, Z., Hong, M., and Niu, M., Appl. Organomet. Chem., 2018, vol. 32, p. e3973. https://doi.org/10.1002/aoc.3973
Shaheen, F., Sirajuddin, M., Ali, S., Dyson, P.J., Shah, N.A., and Tahir, M.N., J. Organomet. Chem., 2018, vol. 856, p. 13. https://doi.org/10.1016/j.jorganchem.2017.12.010
Chans, G.M., Muñoz-Hurtado, J., Hernández-Ortega, S., Ramírez-Apan, T., Nieto-Camacho, A., and Gómez, E., J. Coord. Chem., 2015, vol. 68, p. 3741. https://doi.org/10.1080/00958972.2015.1072174
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
No conflict of interest was declared by the authors.
Rights and permissions
About this article
Cite this article
Shaheen, F., Riaz, L., Naseer, S. et al. Comparative Analysis of Structure and Biopotency of Di- and Triorganotin(IV) Carboxylates with Hexyl Backbone. Russ J Gen Chem 92, 2370–2378 (2022). https://doi.org/10.1134/S1070363222110226
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363222110226