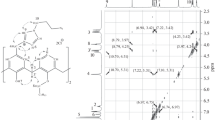
Abstract
The reactions of organosilicon derivatives of octacis(2-hydroxyethylated) calix[4]resorcinarenes with phosphoryl trichloride and subsequent hydrolysis of the resulting phosphorochloridates has afforded novel water-soluble calix[4]resorcinarenes containing eight phosphorus acid fragments at the outer rim of the molecule. The synthesized phosphorodichloridate derivatives on a calixarene platform are convenient precursors for the synthesis of the corresponding phosphoramidates.


Similar content being viewed by others
REFERENCES
Hua, Y., Lan, C., Chenxi, H., Shengbo, L., Zhichao, P., and Yuchao, L., Int. J. Nanomedicine, 2020, vol. 15, p. 5873. https://doi.org/10.2147/ijn.s255637
Supramolecular Amphiphiles, Zhang, X., Ed., Croydon: Royal Society of Chemistry, 2017, p. 171. https://doi.org/10.1039/9781788010566
Thota, B.N.S., Urner, L.H., and Haag, R., Chem. Rev., 2016, vol. 116, p. 2079. https://doi.org/10.1021/acs.chemrev.5b00417
Zhou, Y., Jie, K., and Yao, Y., New J. Chem., 2017, vol. 41, p. 916. https://doi.org/10.1039/C6NJ03026B
Bussolati, R., Carrieri, P., Secchi, A., Arduini, A., Credi, A., Semeraro, M., and Fontana, A., Org. Biomol. Chem., 2013, vol. 11, p. 5944. https://doi.org/10.1039/c3ob40739j
Zhu, H., Shangguan, L., Shi, B., Yu, G., and Huang, F., Mater. Chem. Front., 2018, vol. 2, p. 2152. https://doi.org/10.1039/C8QM00314A
Ji, X., Yao, Y., Li, J., Yan, X., and Huang, F., J. Am. Chem. Soc., 2013, vol. 135, p. 74. https://doi.org/10.1021/ja3108559
Shaban, A. and Eddaif, L., Electroanalysis, 2021, vol. 33, p. 336. https://doi.org/10.1002/elan.202060331
Cretu, C., Maiuolo, L., Lombardo, D., Szerb, E.I., and Calandra, P., J. Nanomaterials, 2020. Article ID 5395048. https://doi.org/10.1155/2020/5395048
Yu, G., Jie, K., and Huang, F., Chem. Rev., 2015, vol. 115, p. 7240. https://doi.org/10.1021/cr5005315
Shah, M.R., Ali, I., Ali, H.S., El-Haj, B.M., and Imran, M., Patent US 10751285 B1, 2020.
Zheng, Z., Geng, W.-C., Xu, Z., and Guo, D.-S., Isr. J. Chem., 2019, vol. 59, p. 913. https://doi.org/10.1002/ijch.201900032
Elliott, T.S., Slowey, A., Yulin Yeb, Y., and Conway, S.J., Med. Chem. Commun., 2012, vol. 3, p. 735. https://doi.org/10.1039/c2md20079a
Wiemer, A.J. and Wiemer, D.F., Top Curr. Chem., 2015, vol. 360, p. 115. https://doi.org/10.1007/128_2014_561
Tian, H.-W., Liu, Y.-C., and Guo, D.-S., Mater. Chem. Front., 2020, vol. 4, p. 46. https://doi.org/10.1039/c9qm00489k
James, E., Eggers, P.K., Harvey, A.R., Dunlop, S.A., Fitzgerald, M., Stubbs, K.A., and Raston, C.L., Org. Biomol. Chem., 2013, vol. 11, p. 6108. https://doi.org/10.1039/c3ob41178h
Fujii, S., Nishina, K., Yamada, S., Mochizuki, S., Ohta, N., Takahara, A., and Sakurai, K., Soft Matter., 2014, vol. 10, p. 8216. https://doi.org/10.1039/c4sm01355g
Lugovskoy, E.V., Gritsenko, P.G., Koshel, T.A., Koliesnik, I.O., Cherenok, S.O., Kalchenko, O.I., Kalchenko, V.I., and Komisarenko, S.V., FEBS J., 2011, vol. 278, p. 1244. https://doi.org/10.1111/j.1742-4658.2011.08045.x
Vovk, A.I., Kalchenko, V.I., Cherenok, S.A., Kukhar, V.P., Muzychka, O.V., and Lozynsky, M.O., Org. Biomol. Chem., 2004, vol. 2, no. 21, p. 3162. https://doi.org/10.1039/B409526J
Trush, V.V., Kharchenko, S.G., Tanchuk, V.Y., Kalchenko, V.I., and Vovk, A.I., Org. Biomol. Chem., 2015, vol. 13, p. 8803. https://doi.org/10.1039/c5ob01247c
Humbert, N., Kovalenko, L., Saladini, F., Giannini, A., Pires, M., Botzanowski, T., Cherenok, S., Boudier, C., Sharma, K.K., Real, E., Zaporozhets, O.A., Cianferani, S., Seguin-Devaux, C., Poggialini, F., Botta, M., Zazzi, M., Kalchenko, V.I., Mori, M., and Mely, Y., ACS Infect. Dis., 2020, vol. 6, p. 687. https://doi.org/10.1021/acsinfecdis.9b00290
Pashirova, T.N., Gibadullina, E.M., Burilov, A.R., Kashapov, R.R., Zhiltsova, E.P., Syakaev, V.V., Habicher, W.D., Rümmeli, M.H., Latypov, Sh.K., Zakharova, L.Ya., and Konovalov, A.I., RSC Adv., 2014, vol. 4, p. 9912. https://doi.org/10.1039/C3RA46146G
McGuigan, C., Bourdin, C., Derudas, M., Hamon, N., Hinsinger, K., Kandil, S., Madela, K., Meneghesso, S., Pertusati, F., Serpi, M., Slusarczyk, M., Chamberlain, S., Kolykhalov, A., Vernachio, J., Vanpouille, C., Introini, A., Margolis, L., and Balzarin, J., Eur. J. Med. Chem., 2013, vol. 70, p. 326. https://doi.org/10.1016/j.ejmech.2013.09.047
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
No conflict of interest was declared by the authors.
Additional information
Translated from Zhurnal Obshchei Khimii, 2021, Vol. 91, No. 10, pp. 1601–1608 https://doi.org/10.31857/S0044460X21100176.
Rights and permissions
About this article
Cite this article
Gibadullina, E.M., Mukhamet’yanova, A.D., Kaupov, A.R. et al. Octacis(2-hydroxyethylated) Calix[4]resorcinarenes Phosphorochloridates as Precursors in Production of Water-Soluble Calix[4]resorcinarene and Phosphoramidates. Russ J Gen Chem 91, 2038–2044 (2021). https://doi.org/10.1134/S1070363221100170
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363221100170