Abstract
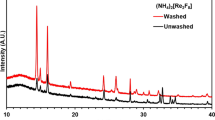
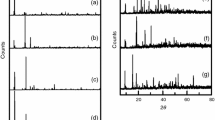
A low-temperature ionic liquid, N-decylpyridinium tetrachloroferrate(III), has been synthesized, and its thermal stability in air has been studied by thermogravimetry. Combustion of the organic part of this ionic liquid leaves a solid residue which has been studied by elemental analysis, X-ray powder diffraction, scanning electron microscopy, IR spectroscopy, and magnetometry. The incombustible residue has been identified as hematite (crystalline α-Fe2O3) which is a weak ferromagnetic at room temperature.
Similar content being viewed by others
References
Vygodskii, Ya.S., Lozinskaya, E.I., and Shaplov, A.S., Ros. Khim. Zh., 2004, vol. 68, no. 6, p. 40.
Gordon, S., Appl. Catal., 2001, vol. 222, p. 101. DOI: 10.1016/S0926-860X.
Zhao, D., Wu, M., Kou, Y., and Min, E., Catal. Today, 2002, vol. 74, p. 157. DOI: 10.1155/2014/729842.
Sun, X. and Zhao, S., Chin. J. Chem. Eng., 2006, vol. 14, p. 289. DOI: 10.1016/S1004-9541(06)60073-6.
Yong, W. and Hong, Y., Chem. Eng. J., 2009, vol. 147, p. 71. DOI: 10.1016/j.cej.2008.11.043.
Xiaowei, P., Ye, H.Y., Liangyu, Y., Pan, Y., Junling, W., Rong, X., and Mary, B.Ch.-P., Carbon, 2010, vol. 48, p. 2501. DOI: 10.1016/j.carbon.2010.03.023.
Liang, L., Yu, H., Guoping, Y., Fangjun, L., Zhiliang, H., and Zhibin, M., Mater. Lett., 2009, vol. 63, p. 8. DOI: 10.1016/j.matlet.2008.08.016.
Rezaei, B., Mallakpour, S., and Taki, M., J. Power Sources, 2009, vol. 187, p. 605. DOI: 10.4061/2011/787905.
Borgel, V., Markevich, E., Aurbach, D., Semrau, G., and Schmidt, M., J. Power Sources, 2009, vol. 189, p. 331. DOI: 10.1016/j.jpowsour.2008.08.099.
Di, W. and Tin, W.N., Electrochem. Commun., 2009, vol. 11, p. 1996. DOI: 10.1016/j.elecom.2009.08.037.
Hayashi, S. and Hamagushi, H., Chem. Lett., 2004, vol. 33, p. 1590. DOI: 10.1002/chin.200518200.
Zhuravlev, O.E., Verolainen, N.V., and Voronchikhina, L.I., Russ. J. Appl. Chem., 2011. vol. 84, no. 7, p. 1158. DOI: 10.1134/S1070427211070068.
Zhuravlev, O.E., Nikol’skii, V.M., and Voronchikhina, L.I., Russ. J. Gen. Chem., 2014, vol. 84, no. 6, p. 1141. DOI: 10.1134/S1070363214060152.
Wyrzykowski, D., Maniecki, T., Pattek-Janczyk, A., Stanek, J., and Warnke, Z., Thermochim. Acta, 2005, vol. 435, p. 92. DOI: 10.1016/j.tca.2005.05.007.
Wyrzykowski, D., Pattek-Janczyk, A., Maniecki, T., Zaremba, K., and Warnke, Z., Thermochim. Acta, 2006, vol. 443, p. 72. DOI: 10.1016/j.tca.2005.12.023.
Schwertmann, U. and Cornell, R.M., The Iron Oxides: Structure, Properties, Reactions, Occurrences and Uses, Weinheim: Wiley-VCH, 2003.
Morish, A.H., Johnston, G.B., and Curry, N.A., Phys. Lett., 1963, vol. 7, p. 177. DOI: 10.1016/0031-9163(63)90372-X.
Artman, J.O., Murphy, J.C., and Foner, S., Phys. Rev., 1965, vol. 138, p. 912. DOI: 10.1103/PhysRev.138.A912.
Pushcharovskii, D.Yu., Rentgenografiya mineralov (X-Ray Diffraction of Minerals), Moscow: Geoinformmark, 2000.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © O.E. Zhuravlev, A.Yu. Karpenkov, D.Yu. Karpenkov, L.I. Voronchikhina, 2015, published in Zhurnal Obshchei Khimii, 2015, Vol. 85, No. 4, pp. 641–647.
Rights and permissions
About this article
Cite this article
Zhuravlev, O.E., Karpenkov, A.Y., Karpenkov, D.Y. et al. Composition and structure of the incombustible residue from thermal decomposition of the ionic liquid N-decylpyridinium tetrachloroferrate(III). Russ J Gen Chem 85, 882–888 (2015). https://doi.org/10.1134/S1070363215040209
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363215040209