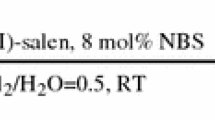Abstract
An efficient catalytic system salen-Mn(III)/NBS for oxidation of secondary alcohols to ketones by inexpensive and readily available oxidizing agent NaOCl has been developed. The process resulted in good to excellent yields under the action of 2 mol % of salen-Mn(III) and 13 mol % of NBS at room temperature. However, such system was not efficient in oxidation of secondary benzyl alcohols with a strong electronicdonating substituent attached to the benzene ring due to bromination of the alcohols.
Similar content being viewed by others
References
Hudlicky, M., Oxidation in Organic Chemistry (ACS Monograph Series), American Chemical Society, Washington, DC: Springer, 1990.
Tojo, G. and Fernádes, M., in Basic Reactions in Organic Synthesis, Tojo, G., Ed., New York: Science + Business Media, Inc. Springer, 2006.
Semmelhack, F.M., Schmidt, C.R., Cortés, D.A., and Chou, C.S., J. Am. Chem. Soc., 1984, vol. 106, p. 3374. DOI: 10.1021/ja00323a064
Dijksman, A., Marino-Gonzalez, A., Mairata i Payeras, A., Arends, I.W.C.E., and Sheldon, R.A., J. Am. Chem. Soc., 2001, vol. 123, p. 6826. DOI: 10.1021/ja0103804
Gamez, P., Arends, I.W.C.E., Reedijk, J., and Sheldon, R.A., Chem. Commun., 2003, p. 2414. DOI: 10.1039/B308668B
Jiang, N. and Ragauskas, A.J., Org. Lett., 2005, vol. 7, p. 3689. DOI: 10.1021/ol051293+
Wang, N.W., Liu, R.H., Chen, J.P., and Liang, X.M., Chem. Commun., 2005, p. 5322. DOI: 10.1039/B509167E
Yian, W.L., Chu, C.H., Lu, Q.Q., Tao, J.W., Liang, X.M., and Liu, R.H., Adv. Synth. Catal., 2010, vol. 352, p. 113. DOI: 10.1002/adsc.200900662
Hoover, J.M. and Stahl, S.S., J. Am. Chem. Soc., 2011, vol. 133, p. 16901. DOI: 10.1021/ja00323a064
Liu, R.H., Liang, X.M., Dong, C.Y., and Hu, X.Q., J. Am. Chem. Soc., 2004, vol. 126, p. 4112. DOI: 10.1021/ja031765k
Xie, Y., Mo, W.M., Xu, D., Shen, Z.L., Sun, N., Hu, B.X., and Hu, X.Q., J. Org. Chem., 2007, vol. 72, p. 4288. DOI: 10.1021/jo0705824
Wang, X.L., Liu, R.H., Jin, Y., and Liang, X.M., Chem. Eur. J., 2008, vol. 14, p. 2679. DOI: 10.1002/chem.200701818
He, X.J., Shen, Z.L., Mo, W.M., Sun, N., Hu, B.X., and Hu, X.Q., Adv. Synth. Catal., 2009, vol. 351, p. 89. DOI: 10.1002/adsc.200800501
Miao, C.X., He, L.N., Wang, J.Q. and Wang, J.L., Adv. Synth. Catal., 2009, vol. 351, p. 2209. DOI: 10.1002/adsc.200900285
Wertz, S. and Studer, A. Adv. Synth. Catal., 2011, vol. 353, p. 69. DOI: 10.1002/adsc.201000703
Wang, L.Y., Li, J., Lv, Y. Zhao, G.D., and Gao, S., Appl. Organomet. Chem., 2012, vol. 26, p. 37. DOI: 10.1002/aoc.1861
Prebil, R., Stavber, G., and Stavber, S., Eur. J. Org. Chem., 2014, p. 395. DOI: 10.1002/ejoc.201301271
Adam, W., Saha-Moller, C.R., and Ganeshpure, P.A., Chem. Rev., 2001, vol. 101, p. 3499. DOI: 10.1021/cr000019k
Sheldon, R.A., Arends, I.W.C.E., ten Brink, G.-J., and Dijksman, A., Acc. Chem. Res., 2002, vol. 35, p. 774. DOI: 10.1021/ar010075n
Parmeggiani, C. and Cardona, F., Green Chem., 2012, vol. 14, p. 547. DOI: 10.1039/C2GC16344F.
Anelli, P.L., Biffi, C., Montanari, F., and Quici, S., J. Org. Chem., 1987, vol. 52, p. 2559. DOI: 10.1021/jo00388a038
Anelli, P.L., Banfi, S., Montanari, F., and Quici, S., J. Org. Chem., 1989, vol. 54, p. 2970. DOI: 10.1021/jo00273a038.
Wolfe, S., Hasan, S.K., and Campbell, J.R., J. Chem. Soc. D, 1970, p.1420. DOI: 10.1039/C29700001420.
Stevens, R.V., Chapman, K.T., and Weller, H.N., J. Org. Chem., 1980, vol. 45, p. 2030. DOI: 10.1021/jo01298a066.
Hu, Z. and Kerton, F.M., Org. Biomol. Chem., 2012, vol. 10, p. 1618. DOI: 10.1039/C2OB06670J
Wang, L., Li, J., Zhao, X., Lv, Y., Zhang, H., and Gao, S., Tetrahedron, 2013, vol. 69, p. 6041. DOI: 10.1016/j.tet.2013.05.076
Ciriminna, R. and Pagliaro, M., Org. Pro. Res. Dev., 2010, vol. 14, p. 245. DOI: 10.1021/op900059x.
Figiel, P.J., Kirillov, A.M., Karabach, Y.Y., Kopylovich, M.N., and Pombeiro, A.J.L., J. Mol. Catal. A: Chem., 2009, vol. 305, p. 178. DOI: 10.1016/j.molcata.2009.01.002
Das, O. and Paine, T.K., Dalton Trans., 2012, vol. 41, p.11476. DOI: 10.1039/C2DT31134H
Kuang, Y., Nabae, Y., Hayakawa, T., and Kakimoto, M. Green Chem., 2011, vol. 13, p. 1659. DOI: 10.1039/C1GC15076F.
Sun, W., Wang, H., Xia, C., Li, J., and Zhao, P., Angew. Chem. Int. Ed., 2003, vol. 42, p. 1042. DOI: 10.1002/anie.200390268
Li, Z., Tang, Z.H., Hu, X.X., and Xia, C.G., Chem. Eur. J., 2005, vol. 11, p. 1210. DOI: 10.1002/chem.200400818
Sun, W., Wu, X., and Xia, C., Helv. Chim. Acta, 2007, vol. 90, p. 623. DOI: 10.1002/hlca.200790064
Cheng, Q., Deng, F., Xia, C., and Sun, W., Tetrahedron: Asymmetry, 2008, vol. 19, p. 2359. DOI: 10.1016/j.tetasy.2008.10.008.
Brown, M.K., Blewett, M.M., Colombe, J.R., Corey, E.J., J. Am. Chem. Soc., 2010, vol. 132, p. 11165. DOI: 10.1021/ja103103d.
Xu, D., Wang, S., Shen, Z., Xia, C.G., and Sun, W., Org. Biomol. Chem., 2012, vol. 10, p. 2730. DOI: 10.1039/C2OB07087A.
Zhang, Y.C., Zhou, Q., Ma, W.C., and Zhao, J.Q., Catal. Commun., 2014, vol. 45, p. 114. DOI: 10.1016/j.catcom.2013.11.007.
Larrow, J.F. and Jacobsen, E.N., J. Org. Chem., 1994, vol. 59, p. 1939. DOI: 10.1021/jo00086a062.
Pääkkönen, S., Pursiainen, J., and Lajunen, M., Synth. Commun., 2012, vol. 42, p. 534. DOI: 10.1080/00397911.2010.526283.
Krishnaveni, N.S., Surendra, K., and Rao, K.R., Adv. Synth. Catal., 2004, vol. 346, p. 346. DOI: 10.1002/adsc.200303164
Venkatasubramanian, N. and Thiagarajan, V., Tetrahedron Lett., 1968, vol. 14, p. 1711.
Venkatasubramanian, N. and Thiagarajan, V., Can. J. Chem., 1969, vol. 47, p. 694. DOI: 10.1139/v69-108.
Karimi, B. and Badreh, E., Org. Biomol. Chem., 2011, vol. 9, p. 4194. DOI: 10.1039/C1OB05178D.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Zhang, Y.C., Lü, F.L., Cui, M.J. et al. Efficient oxidation of secondary alcohols to Ketones by NaOCl catalyzed by Salen-Mn(III)/NBS. Russ J Gen Chem 84, 2021–2026 (2014). https://doi.org/10.1134/S1070363214100272
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363214100272