Abstract
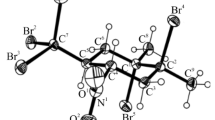
3,3,3-Tribromo-1-nitropropene and its precursors such as 2-hydroxy- and 2-acetoxy-1,1,1-tribromo-1-nitropropanes were prepared and characterized by IR, UV, 1H, 13C, HMQC, and HMBC NMR spectroscopy methods. Structure of tribromomethylnitroalkene was studied by X-ray diffraction.
Similar content being viewed by others
References
Korotaev, V.Yu., Barkov, A.Yu., Sosnovskikh, V.Ya., Slepukhin, P.A., and Kodess, M.I., Tetrahedron Lett., 2011, vol. 52, no. 44, p. 5764. DOI: 10.1016/j.tetlet. 2011.08.109.
Korotaev, V.Yu., Sosnovskikh, V.Ya., Barabanov, M.A., Yasnova, E.S., Ezhikova, M.A., Kodess, M.I., and Slepukhin, P.A., Tetrahedron, 2010, vol. 66, no. 6, p. 1404. DOI: 10.1016/j.tet.2009.11.094.
Berestovitskaya, V.M., Makarenko, S.V., Bushmarinov, I.S., Lyssenko, K.A., Smirnov, A.S., and Stukan’, E.V., Russ. Chem. Bull., 2009, vol. 58, no. 5, p. 1023. DOI: 10.1007/s11172-009-0131-2.
Shechter, H., Ley, D.E., and Roberson, E.B., J. Am. Chem. Soc., 1956, vol. 78, p. 4984. DOI: 10.1021/ja01600a049.
Irving, H. and Fuller, H.I., J. Chem. Soc., 1948, p. 1989. DOI: 10.1039/JR9480001987.
Brower, F. and Burkett, H., J. Am. Chem. Soc., 1953, vol. 75, no. 5, p. 1082. DOI: 10.1021/ja01101a021.
Ivanova, I.S., Sadykov, R.A., Petukhov, V.A., and Gudovich, L.P., Bull. Acad. Sci. of the USSR, Chem. Sci., 1968, vol. 17, no. 9, p. 2038. DOI: 10.1007/BF00905017.
Bates, A.N., Spencer, D.M., and Wain, R.L., Ann. Appl. Biol., 1963, vol. 51, no. 1, p. 153. DOI: 10.1111/j.1744-7348.1963.tb03682.x.
Bluestone, H. and Heights, C., US Patent 2917551, 1959, C. A., 1959, vol. 53, p. 22718a.
Molteni, M., Volonterio, A., and Zanda, M., Org. Lett., 2003, vol. 5, no. 21, p. 3887. DOI: 10.1021/ol0354730.
Molteni, M., Bigotti, S., Volonterio, A., Bellucci, M.C., Mazzini, S., and Zanda, M., Org. Biomol. Chem., 2009, vol. 7, no. 11, p. 2286. DOI: 10.1039/B901718F.
Korotaev, V.Yu., Kutyashev, I.B., Barkov, A.Yu., Kodess, M.I., Slepukhin, P.A., and Zapevalov, A.Ya., Russ. Chem. Bull., 2009. Vol. 58, no. 9, p. 1886. DOI: 10.1007/s11172-009-0257-2.
Turconi, J., Lebeau, L., Paris, J.-M., and Mioskowski, C., Tetrahedron Lett., 2006, vol. 47, no. 1, p. 121. DOI: 10.1016/j.tetlet.2005.10.116.
Slobodchikova, E.K., Anisimova, N.A., and Berestovitskaya, V.M., Russ. J. Gen. Chem., 2013, vol. 83, no. 8, p. 1631. DOI: 10.1134/S107036321308029X.
Korotaev, V.Yu., Sosnovskikh, V.Ya., Barkov, A.Yu., Slepukhin, P.A., Ezhikova, M.A., Kodess, M.I., and Shklyaev, Y.V., Tetrahedron, 2011, vol. 67, no. 45, p. 8685. DOI: 10.1016/j.tet.2011.09.049.
Korotaev, V.Yu., Sosnovskikh, V.Ya., Kutyashev, I.B., Barkov, A.Yu., Matochkina, E.G., and Kodess, M.I., Tetrahedron, 2008, vol. 64, no. 22, p. 5055. DOI: 10.1016/j.tet.2008.03.065.
Bigotti, S., Malpezzi, L., Molteni, M., Mele, A., Panzeri, W., and Zanda, M., Tetrahedron Lett., 2009, vol. 50, no. 21, p. 2540. DOI: 10.1016/j.tetlet.2009.03.059.
Balthazor, T.M., Gaede, B., Korte, D.E., and Shieh, H.-S., J. Org. Chem., 1984, vol. 49, no. 23, p. 4547. DOI: 10.1021/jo00197a049.
Anisimova, N.A., Slobodchikova, E.K., Berestovitskaya, V.M., Kuzhaeva, A.A., Ivanova, M.E., and Rybalova, T.V., Russ. J. Gen. Chem., 2014, vol. 84, no. 2, p. 242. DOI: 10.1134/S1070363214020145
Ishmaeva, E.A., Gazizova, A.A., Vereshchagina Ya.A., Chachkov, D.V., Anisimova, N.A., Makarenko, S.V., Smirnov, A.S., and Berestovitskaya, V.M., Russ. J. Gen. Chem., 2007, vol. 77, no. 5, p. 894. DOI: 10.1134/S1070363207050131.
Arikava, Y., Dong, Q., Feher, V., and Jones, B., US Patent 152273, 2011.
Silverstein, R.M., Webster, F.X., and Kiemle, D., Spectrometric Identification of Organic Compounds, Wiley Global Education, 2005.
Miller, D.B., Flanagan, P.W., and Shechter, H., J. Org. Chem., 1976, vol. 41, no. 12, p. 2112. DOI: 10.1021/jo00874a010.
Tronchet, J.M.J., Zerelli, S., and Bernardinelli, G., J. Carbohydr. Chem., 1999, vol. 18, p. 343. DOI: 10.1080/073283099085 44000.
Allen, F.H., Acta Crystallogr. (B), 2002, vol. 58, p. 380. DOI: 10.1107/S0108768102003890; Bruno, I.J., Cole, J.C., Edgington, P.R., Kessler, M., Macrae, C.F., McCabe, P., Pearson, J., and Taylor, R., Acta Crystallogr. (B), 2002, vol. 58, p. 389. DOI: 10.1107/S0108768102003324.
Rowland, R.S. and Taylor, R., J. Phys. Chem., 1996, vol. 100, p. 7384. DOI: 10.1021/jp953141.
Kostikov, R.R., Kuznetsov, M.S., Novikov, M.S., Sokolov, V.V., and Khlebnikov, A.F., Praktikum po organicheskomu sintezu (Workshop on Organic Synthesis), St. Petersburg:, S.-Peterburg. Gos. Univ., 2009, p. 257.
Chattaway, F.D., Witherington, P., J. Chem. Soc., 1935, p. 1178. DOI: 10.1039/JR9350001178.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.A. Anisimova, E.K. Slobodchikova, A.A. Kuzhaeva, T.V. Rybalova, E.V. Stukan’, V.M. Berestovitskaya, 2014, published in Zhurnal Obshchei Khimii, 2014, Vol. 84, No. 5, pp. 741–745.
Rights and permissions
About this article
Cite this article
Anisimova, N.A., Slobodchikova, E.K., Kuzhaeva, A.A. et al. 3,3,3-Tribromo-1-nitropropene: Synthesis and structure. Russ J Gen Chem 84, 834–838 (2014). https://doi.org/10.1134/S1070363214050089
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363214050089