Abstract
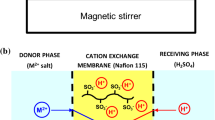
The process of membrane extraction of rare earth ions, Nd(III), Sc(III), and Sm(III), and also the accompanying them in the natural raw material ions Al(III) and Mg(II), by N,N-bis(dihexylphosphorylmethyl)octylamine (BPA) was investigated under the conditions of active membrane transport, and its high efficiency and selectivity was demonstrated with respect to the two first rare earth metals. The influence of principal parameters of the three-phase extraction process, including the nitrate ion concentration, the releasing aqueous phase acidity, the extracting agent concentration in the membrane, and the nature of the membrane solvent, on the membrane permeability, characterizing the efficiency and selectivity of membrane transporters, was established. Comparison of the membrane permeability in the process of transmembrane transport of Nd(III) and Sc(III) with BPA and monophosphorylated amines showed a significant advantage of the biphosphorylated amine extractant.
Similar content being viewed by others
References
Cherkasov, R.A., Garifzyanov, A.R., Galeev, R.R., Kurnosova, N.V., Davletshin, R.R., Zakharov, S.V., Zh. Obshch. Khim., 2011, vol. 81, no. 7, p. 1114.
Cherkasov, R.A., Garifzyanov, A.R., Leont’eva, S.V., Davletshin, R.R., and Koshkin, S.A., Zh. Obshch. Khim., 2009, vol. 79, no. 12, p. 1973.
Mulder, M., Basic Principles of Membrane Technology, New York: Springer, 1991.
Membrane Separation Technology: Principles and Applications, Noble, E.D. and Stern, S.T., Eds., Amsterdam: Elsevier, 1995.
Cherkasov, R.A., Garifzyanov, A.R., Bazanova, E.B., Davletshin, R.R., and Leont’eva, S.V., Zh. Obshch. Khim., 2012, vol. 82, no. 1, p. 36.
Garifzyanov, A.R., Davletshin, R.R., Davletshina, N.V., and Cherkasov, R.A., Zh. Obshch. Khim., 2012, vol. 82.
Ivakhno, S.Yu., Yagodin, G.A., and Afanas’ev, A.V., Itogi Nauki i Tehniki, Ser. Neorg. Khim., Moscow: VINITI, 1985, vol. 13.
Garifzyanov, A.R., Davletshin, R.R., Davletshina, N.V., and Cherkasov, R.A., Zh. Obshch. Khim., 2012, vol. 82.
Wang, W., Pranolo, Y., and Cheng, C., Hydrometallurgy, 2011, vol. 108, nos. 1–2, p. 100.
Cherkasov, R.A., Garifzyanov, A.R., Bazanova, O.B., and Leont’eva, S.V., Zh. Obshch. Khim., 2011, vol. 81, no. 10, p. 1627.
Garifzyanov, A.R., Nuriazdanova, G.Kh., Zakharov, S.V., and Cherkasov, R.A., Zh. Obshch. Khim., 2004, vol. 74, no. 12, p. 1998.
Cherkasov, R.A., Garifzyanov, A.R., Krasnova, N.S., Cherkasov, A.R., and Talan, A.S., Zh. Obshch. Khim., 2006, vol. 76, no. 10, p. 1603.
Karyakin, Yu.V. and Angelov, I.I., Chistye himicheskie veshhestva (Pure Chemical Compounds), Moscow: Khimiya, 1974.
Cherkasov, R.A., Garifzyanov, A.R., Talan, A.S., Davletshin, R.R., and Kurnosova, N.V., Zh. Obshch. Khim., 2009, vol. 79, no. 9, p. 1480.
Tikhonov, V.N., Analiticheskaya khimiya alyuminiya (Analytical Chemistry of Aluminum), Moscow: Nauka, 1971.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.R. Garifzyanov, N.V. Davletshina, E.Yu. Myatish, R.A. Cherkasov, 2013, published in Zhurnal Obshchei Khimii, 2013, Vol. 83, No. 2, pp. 213–219.
Rights and permissions
About this article
Cite this article
Garifzyanov, A.R., Davletshina, N.V., Myatish, E.Y. et al. Membrane extraction of metal ions by aminophosphoryl reagents in the active transport conditions. Russ J Gen Chem 83, 267–273 (2013). https://doi.org/10.1134/S1070363213020060
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363213020060