Abstract
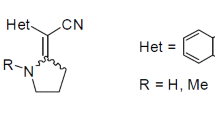
Reactions of N-alkyl-2-chloropyridinium salts with benzimidazolylacetonitriles result in (E)-2-(1Hbenzo[d]imidazol-2-yl)-2-[1-alkylpyridin-2(1H)-ylidene]acetonitriles. The alkylation of the latter with ω-bromoacetophenones in boiling acetone may gives rise to the N-alkylated salts, which are stabilized in two configurations, Z and E. The heating of the salts in acetonitrile causes their transformation into 2-(1H-benzo[d]-imidazol-2(3H)-ylidene(cyano)methyl)-1-methylpyridinium bromide due to the dearoylmethylation. The structure of the latter was proved by the XRD analysis.
Similar content being viewed by others
References
Khoroshilov, G., Demchak, I., and Saraeva, T., Synthesis, 2008, no. 10, p. 1541.
Mukaiyama, T., Angew. Chem. Int. Ed., 1979, vol. 18, no. 10, p. 707.
Mukaiyama, T., Challenges in synthetic organic chemistry, Oxford: Oxford Univ. Press, 2002, 226 p.
Zefirov, Yu.V. and Zorkii, P.M., Usp. Khim., 1989, vol. 58, no. 5, p. 713.
Burgi, H.-B. and Dunitz, J.D., Structure correlation, Weinheim: VCH, 1994, vol. 2, p. 741.
Sheldrick, G., Acta Cryst. (A), 2008, vol. 64, no. 1, p. 112.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © T.A. Saraeva, G.E. Khoroshilov, R.I. Zubatyuk, O.V. Shishkin, 2012, published in Zhurnal Obshchei Khimii, 2012, Vol. 82, No. 4, pp. 659–664.
Rights and permissions
About this article
Cite this article
Saraeva, T.A., Khoroshilov, G.E., Zubatyuk, R.I. et al. Mukaiyama reagents in the synthesis of (E)-2-(1H-benzo[d]imidazol-2-yl)-2-[1-alkylpyridin-2(1H)ylidene]acetonitriles and their further electronic rearrangements effected by the action of acids and alkylating agents. Russ J Gen Chem 82, 744–748 (2012). https://doi.org/10.1134/S1070363212040238
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363212040238