Abstract
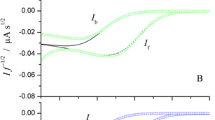
The processes of electrochemical reduction of Ti(IV) and oxidation of Ti(III) in aqueous solutions of H2SO4 not containing and containing AcOH or MeCN are studied by the methods of classical and commutation polarography. It is shown that in the absence of organic solvents the heterogenous electron transfer is irreversible in the media with the sulfuric acid concentration up to 9 M, while in aqueous-organic solution the same occurs at the concentration up to 7 M. Organic solvents are involved into the process of complex formation and like H2SO4, influence the step of heterogenous electron transfer; this effects weakens with the increase in the concentration of sulfuric acid.
Similar content being viewed by others
References
Noel, M., Anantharaman, P.N., and Udupa, H.V.K., J. Appl. Electrochem., 1982, no.12, p. 291.
Udupa, H.V.K., Indian. Chem. Eng., 1988, vol. 30, no.1, p. 53.
Gul’tyai, V.P. and Leibzon, V.N., Elektrokhimiya, 1996, vol. 32, no.1, p. 65.
Lisitsyn, Yu.A. and Kargin, Yu.M., Zh. Obshch. Khim., 1996, vol. 66, no.1, p. 89.
Lisitsyn, Yu.A. and Kargin, Yu.M., Elektrokhimiya, 2000, vol. 36, no.2, p. 103.
Lisitsyn, Yu.A., Busygina, N.V., and Kargin, Yu.M., Rus. Khim. Zh., 2005, vol. 49, no.5, p. 121.
Lisitsyn, Yu.A. and Kargin, Yu.M., Elektrokhimiya, 2004, vol. 40, no.9, p. 1129.
Lisitsyn, Yu.A. and Kargin, Yu.M., Zh. Obshch. Khim., 1993, vol. 63, no.6, p. 1312.
Godneva, M.M. and Motov, D.A., Khimiya podgruppy titana: Sul’faty i ikh tastvory (Chemistry of Titanium Subgroup: Sulfates and Their Solutions), Leningrad: Nauka, 1980.
Fatouros, N., Krulie, D., and Larabi, N., J. Electroanal. Chem., 2004, vol. 568, p. 55.
Organicheskaya elektrokhimiya (Organic Electro-chemistry), Beizer, M. and Lund, K., Eds., Moscow: Khimiya, 1988.
Habashy, G.M., Collect. Czech. Chem. Commun., 1960, vol. 25, no.12, p. 3166.
Habashy, G.M., Z. Anorg. Allgem. Chem., 1960, vol. 306, nos. 5–6, p. 312.
Bishop, E. and Hitchcock, P.H., Analyst, 1973, vol. 98, no.1170, p. 625.
Noel, M., Srinivasan, R.K., and Chidambaran, S., Bull. Electrochem., 1986, vol. 2, no.4, p. 413.
Kargin, Yu.M. and Latypova, V.Z., Itogi nauki and tekhniki: Fizicheskaya khimiya, elektrokhimiya (Summary of Science and Technology: Physical Chemistry, Electrochemistry), Moscow: VINITI, 1981, vol. 7, p. 210.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © Yu.A. Lisitsyn, N.V. Busugina, 2009, published in Zhurnal Obshchei Khimii, 2009, Vol. 79, No.7, pp. 1057–1064.
Rights and permissions
About this article
Cite this article
Lisitsyn, Y.A., Busugina, N.V. Electrochemical behavior of Ti(IV)/Ti(III) redox couple on dropping mercury electrode in sulfuric acid aqueous and aqueous-organic media. Russ J Gen Chem 79, 1413–1420 (2009). https://doi.org/10.1134/S1070363209070019
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363209070019