Abstract
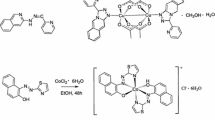
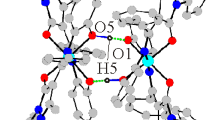
β-Aminovinyl ketone (2-nitro-3-(8-quinolylamino)prop-2-enal) was synthesized by condensation of nitromalondialdehyde with 8-aminoquinoline. The reactions of β-aminovinyl ketone with copper, nickel, and cobalt acetates in methanol gave metal complexes. The spectral and magnetic characteristics of preparatively isolated compounds were studied. The structure of cobalt(II) chelate was determined on the basis of X-ray diffraction study (CIF file CCDC no. 2109263).


Similar content being viewed by others
REFERENCES
Bourget-Merle, L., Lappert, M.F., and Severn, J.R., Chem. Rev., 2002, vol. 102, no. 9, p. 3031. https://doi.org/10.1021/cr010424r
Camp, C. and Arnold, J., Dalton Trans., 2016, vol. 45, no. 37, p. 14462. https://doi.org/10.1039/C6DT02013E
Puring, K., Zywitzki, D., Taffa, D.H., et al., Inorg. Chem., 2018, vol. 57, no. 9, p. 5133. https://doi.org/10.1021/acs.inorgchem.8b00204
Lyubov, D.M., Tolpygin, A.O., and Trifonov, A.A., Coord. Chem. Rev., 2019, vol. 392, p. 83. https://doi.org/10.1016/j.ccr.2019.04.013
Witkowska, E., Orwat, B., Oh, M.J., et al., Inorg. Chem., 2019, vol. 58, no. 22, p. 1567. https://doi.org/10.1021/acs.inorgchem.9b02785
Huster, N., Zanders, D., Karle, S., et al., Dalton Trans., 2020, vol. 49, no. 31, p. 10755.https://doi.org/10.1039/d0dt01463J
Zywitzki, D., Taffa, D.H., Lamkowski, L., et al., Inorg. Chem., 2020, vol. 59, no. 14, p. 10059. https://doi.org/10.1021/acs.inorgchem.0c01204
Allison, M., Wilson, D., Pask, C.M., et al., Chembiochem, 2020, vol. 21, no. 14, p. 1988. https://doi.org/10.1002/cbic.202000028
Lord, R.M., Hebden, A.J., Pask, C.M., et al., J. Med. Chem., 2015, vol. 58, no. 12, p. 4940. https://doi.org/10.1021/acs.jmedchem.5b00455
Bermeshev, M.V. and Chapala, P.P., Prog. Polymer Sci., 2018, vol. 84, p. 1. https://doi.org/10.1016/j.progpolymsci.2018.06.003
Yao, S. and Driess, M., Acc. Chem. Res., 2012, vol. 45, no. 2, p. 276. https://doi.org/10.1021/ar200156r
Di Francesco, G.N., Gaillard, A., Ghiviriga, I., et al., Inorg. Chem., 2014, vol. 53, no. 9, p. 4647. https://doi.org/10.1021/ic500333p
Lai, P.-N., Brysacz, C.H., Alam, M.K., et al., J. Am. Chem. Soc., 2018, vol. 140, no. 32, p. 10198. https://doi.org/10.1021/jacs.8b04841
Korshunov, O.Y., Uraev, A.I., Shcherbakov, I.N., et al., Russ. J. Inorg. Chem., 2000, vol. 45, no. 9, p. 1363.
Uraev, A.I., Kurbatov, V.P., Nivorozhkin, A.L., et al., Russ. Chem. Bull. Int. Ed., 2002, vol. 51, no. 10, p. 1924. https://doi.org/10.1023/A:1021321022710
Uraev, A.I., Kurbatov, V.P., Tylchenko, L.S., et al., Dokl. Chem., 2002, vol. 383, no. 1, p. 57.
Uraev, A.I., Ikorskii, V.N., Bubnov, M.P., et al., Russ. J. Coord.Chem., 2006, vol. 32, no. 4, p. 287. https://doi.org/10.1134/S1070328406040105
Fanta, P.E., Organ. Synth., 1952, vol. 32, p. 95. https://doi.org/10.15227/orgsyn.032.0095
Becke, A.D., J. Chem. Phys., 1993, vol. 98, no. 7, p. 5648. https://doi.org/10.1063/1.464913
Frisch, M.J., Trucks, G.W., Schlegel, H.B., et al., Gaussian 09, Revision A.02, 2009.
Zhurko, G.A. and Zhurko, D.A., Chemcraft ver. 1.6 (build 338). http://www.chemcraftprog.com.
Sheldrick, G., Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, vol. 64, no. 1, p. 112. https://doi.org/10.1107/S0108767307043930
Uraev, A.I., Nivorozhkin, A.L., Kurbatov, V.P., et al., Russ. J. Coord. Chem., 2000, vol. 26, no. 12, p. 891. https://doi.org/10.1023/A:1026639327693
Yokota, S., Tachi, Y., Nishiwaki, N., et al., Inorg. Chem., 2001, vol. 40, no. 21, p. 5316. https://doi.org/10.1021/ic0155535
Spencer, D.J.E., Reynolds, A.M., Holland, P.L., et al., Inorg. Chem., 2002, vol. 41, no. 24, p. 6307. https://doi.org/10.1021/ic020369k
Zatka, V., Holzbecher, J., and Ryan, D.E., Anal. Chim. Acta, 1971, vol. 55, no. 1, p. 2738. https://doi.org/10.1016/S0003-2670(01)82767-7
Fritsch, J.M., Thoreson, K.A., and McNeill, K., Dalton Trans., 2006, no. 40, p. 4814.https://doi.org/10.1039/B609616F
Dorovskikh, S.I., Alexeyev, A.V., Kuratieva, N.V., et al., J. Organomet. Chem., 2013, vol. 741, p. 122. https://doi.org/10.1016/j.jorganchem.2013.05.001
Garnovskii, A.D., Nivorozhkin, A.L., and Minkin, V.I., Coord. Chem. Rev., 1993, vol. 126, nos. 1−2, p. 1. https://doi.org/10.1016/0010-8545(93)85032-y
Kotova, O., Lyssenko, K., Rogachev, A., et al., J. Photochem. Photobiol., A, 2011, vol. 218, p. 117. https://doi.org/10.1016/j.jphotochem.2010.12.011
Robson, K.C.D., Phillips, C.D., Patrick, B.O., et al., Dalton Trans., 2010, vol. 39, no. 10, p. 2573. https://doi.org/10.1039/b921153e
Gong, D., Wang, B., Jia, X., et al., Dalton Trans., 2014, vol. 43, no. 10, p. 4169. https://doi.org/10.1039/C3DT52708E
Funding
This study was supported by the Ministry of Science and Higher Education of the Russian Federation (state assignment of the Southern Federal University in the field of science, project 0852-2020-0031).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by Z. Svitanko
Rights and permissions
About this article
Cite this article
Kovaleva, T.V., Uraev, A.I., Lyssenko, K.A. et al. Synthesis, Structure, and Properties of Copper(II), Nickel(II), and Cobalt(II) Ketoiminate Chelates. Molecular and Crystal Structures of Bis[2-nitro-3-(8-quinolylimino)prop-1-enoxy]cobalt(II). Russ J Coord Chem 48, 210–217 (2022). https://doi.org/10.1134/S1070328422040029
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328422040029