Abstract
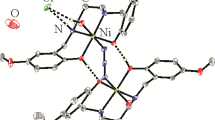
Three nickel(II), manganese(II) and cadmium(II) complexes, [NiL2] (I), [MnL2] (II) and [Cd(HL)2(NO3)]·NO3 (III), derived from 4-bromo-N'-(1-(pyridin-2-yl)ethylidene)benzohydrazide (HL) were prepared and characterized by IR and UV spectra. Structures of the complexes were further confirmed by single crystal X-ray diffraction techniques (CIF files CCDC nos. 1874307 (I), 1874308 (II), and 1874309 (III)). The Ni atom in complex I and the Mn atom in complex II are in octahedral coordination. The Cd atom in complex III is in square antiprism coordination. The nickel complex is effective on the inhibition of Jack bean urease.


Similar content being viewed by others
REFERENCES
Angelusiu, M.V., Barbuceanu, S.F., Draghici, C., et al., Eur. J. Med. Chem., 2010, vol. 45, no. 5, p. 2055.
Ajani, O.O., Obafemi, C.A., Nwinyi, O.C., et al., Bioorg. Med. Chem., 2010, vol. 18, no. 1, p. 214.
Zhang, Y.H., Zhang, L., Liu, L., et al., Inorg. Chim. Acta, 2010, vol. 363, no. 2, p. 289.
Horiuchi, T., Chiba, J., Uoto, K., et al., Bioorg. Med. Chem. Lett., 2009, vol. 19, no. 2, p. 305.
El-Sayed, M.A.A., Abdel-Aziz, N.I., Abdel-Aziz, A.A.M., et al., Bioorg. Med. Chem., 2011, vol. 19, no. 11, p. 3416.
Sondhi, S.M., Dinodia, M., and Kumar, A., Bioorg. Med. Chem., 2006, vol. 14, no. 13, p. 4657.
Krishnamoorthy, P., Sathyadevi, P., Cowley, A.H., et al., Eur. J. Med. Chem., 2011, vol. 46, no. 8, p. 3376.
Avaji, P.G., Kumar, C.H.V., Patil, S.A., et al., Eur. J. Med. Chem., 2009, vol. 44, no. 9, p. 3552.
Khan, K.M., Rahim, F., Khan, A., et al., J. Chem. Soc. Pak., 2015, vol. 37, no. 3, p. 479.
Das, K., Sinha, C., and Datta, A., J. Chem. Crystallogr., 2012, vol. 42, no. 2, p. 150.
Bernhardt, P.V., Caldwell, L.M., Chaston, T.B., et al., J. Biol. Inorg. Chem., 2003, vol. 8, no. 8, p. 866.
Wu, X., Zhang, D., Deng, S., et al., Inorg. Chem. Commun., 2017, vol. 84, p. 195.
Koo, B.-K., J. Korean Chem. Soc., 2013, vol. 57, no. 6, p. 859.
Sathyadevi, P., Krishnamoorthy, P., Alagesan, M., et al., Polyhedron, 2012, vol. 31, no. 1, p. 294.
Ara, R., Ashiq, U., Mahroof-Tahir, M., et al., Chem. Biodiversity, 2007, vol. 4, no. 1, p. 58.
Li, Y.T., Dong, J.W., Lu, Y., et al., Chinese J. Inorg. Chem., 2018, vol. 34, no. 6, p. 1192.
Pan, L., Wang, C., Yan, K., et al., J. Inorg. Biochem., 2016, vol. 159, p. 22.
You, Z., Liu, M., Wang, C., et al., RSC Advances, 2016, vol. 6, no. 20, p. 16679.
Cheng, K., You, Z.L.,and Zhu, H.L., Aust. J. Chem., 2007, vol. 60, no. 5, p. 375.
You, Z.L. and Zhou, P., Inorg. Chem. Commun., 2007, vol. 10, no. 11, p. 1273.
Cheng, X.S., Zhang, J.C., You, Z.L., et al., Transition Met. Chem., 2014, vol. 39, no. 3, p. 291.
Qu, D., Niu, F., Zhao, X.L., et al., Bioorg. Med. Chem., 2015, vol. 23, no. 9, p. 1944.
Zhang, M., Xian, D.M., Li, H.H., et al., Aust. J. Chem., 2012, vol. 65, no. 4, p. 343.
Bruker, SMART (version 5.628) and SAINT (version 6.02), Madison: Bruker AXS, 1998.
Sheldrick, G.M., SADABS, Program for Empirical Absorption Correction of Area Detector, Göttingen: Univ. of Göttingen, 1996.
Sheldrick, G.M., Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, vol. 64, no. 1, p. 112.
Geary, W.J., Coord. Chem. Rev., 1971, vol. 7, no. 1, p. 81.
Datta, A., Chuang, N.-T., Sie, M.-H., et al., Acta Crystallogr., Sect. E: Struct. Rep. Online, 2010, vol. 66, no. 4, p. m359.
Shit, S., Chakraborty, J., Samanta, B., et al., Struct. Chem., 2009, vol. 20, no. 4, p. 633.
Ray, A., Banerjee, S., Sen, S., et al., Struct. Chem., 2008, vol. 19, no. 2, p. 209.
Jang, Y.J., Lee, U., and Koo, B.K., Bull. Korean Chem. Soc., 2005, vol. 26, no. 6, p. 925.
Shen, F., Huang, W., Huang, X., et al., Transition Met. Chem., 2015, vol. 40, no. 6, p. 681.
Sen, S., Talukder, P., Rosair, G., et al., Struct. Chem., 2005, vol. 16, no. 6, p. 605.
Chandra, S. and Sharma, A.K., Spectrochim. Acta, Part A, 2009, vol. 72, no. 4, p. 851.
Shyu, H.-L., Wei, H.-H., and Wang, Y., Inorg. Chim. Acta, 1999, vol. 290, no. 1, p. 8.
Tanaka, T., Kawase, M., and Tani, S., Life Sci., 2003, vol. 73, no. 23, p. 2985.
Funding
This work was supported by the Science and Technology Research Project of Hubei Provincial Department of Education (project no. B2018061), the Hubei Provincial University Teaching Team “Environmental Control and Energy Resource Utilization Teaching Team” (project no. 109), and the Environmental Engineering and Science Major Course Teaching Team of Wuhan Textile University (project no. B2018061).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhao, H., Tan, X.P., Peng, Q.A. et al. Synthesis, Characterization, Crystal Structures, and Biological Activity of Nickel(II), Manganese(II) and Cadmium(II) Complexes Derived from 4-Bromo-N'-(1-(Pyridin-2-YL)ethylidene)benzohydrazide. Russ J Coord Chem 47, 58–65 (2021). https://doi.org/10.1134/S107032842011010X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107032842011010X