Abstract
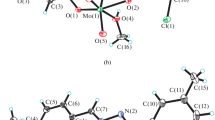
The preparation of Mo(VI) hydrazone complexes, cis-[MoO2L1(CH3OH)] (I) and cis-[MoO2L2(CH3OH)] (II), derived from N'-(3-bromo-2-hydroxybenzylidene)-2-chlorobenzohydrazide (H2L1) and N'-(3-bromo-2-hydroxybenzylidene)-4-bromobenzohydrazide (H2L2), respectively, is reported. The complexes were characterized by elemental analyses, infrared and electronic spectroscopy, and single crystal structure analysis (CIF files ССDС nos. 1426875 (I), 1426871 (II)). The Mo atoms are coordinated by two cis terminal oxygen, ONO from the hydrazone ligand, and methanol oxygen. Even though the hydrazone ligands and the coordination sphere in both complexes are similar, the unit cell dimensions and the space groups are different. Complex I crystallized as orthorhombic space group Pca21 with unit cell dimensions a = 27.887(2), b = 8.0137(7), c = 15.544(1) Å, V = 3473.8(5) Å3, Z = 8, R 1 = 0.0450, wR 2 = 0.0539. Complex II crystallized as triclinic space group P1, with unit cell dimensions a = 8.2124(4), b = 8.5807(5), c = 12.9845(8) Å, α = 83.366(2)°, β = 79.201(2)°, γ = 80.482(2)°, V = 883.03(9) Å3, Z = 2, R 1 = 0.0278, wR 2 = 0.0569. The complexes were tested as catalyst for the oxidation of olefins, and showed effective activity.
Similar content being viewed by others
References
Haque, M.R., Ghosh, S., Hogarth, G., et al., Inorg. Chim. Acta, 2015, vol. 434, p. 150.
Szatkowski, L. and Hall, M.B., Inorg. Chem., 2015, vol. 54, no. 13, p. 6380.
Hatnean, J.A. and Johnson, S.A., Dalton Trans., 2015, vol. 44, no. 33, p. 14925.
Chatterjee, I., Chowdhury, N.S., Ghosh, P., et al., Inorg. Chem., 2015, vol. 54, no. 11, p. 5257.
Sieh, D., Lacy, D.C., Peters, J.C., et al., Chem. Eur. J., 2015, vol. 21, no. 23, p. 8497.
Rayati, S., Rafiee, N., and Wojtczak, A., Inorg. Chim. Acta, 2012, vol. 386, p. 27.
Alemohammad, T., Safari, N., Rayati, S., et al., Inorg. Chim. Acta, 2015, vol. 434, p. 198.
Kissel, A.A., Mahrova, T.V., Lyubov, D.M., et al., Dalton Trans., 2015, vol. 44, no. 27, p. 12137.
Bagherzadeh, M., Ghanbarpour, A., and Khavasi, H.R., Catal. Commun., 2015, vol. 65, p. 72.
Pranckevicius, C., Fan, L., and Stephan, D.W., J. Am. Chem. Soc., 2015, vol. 137, no. 16, p. 5582.
Safin, D.A., Pialat, A., Korobkov, I., et al., Chem. Eur. J., 2015, vol. 21, no. 16, p. 6144.
Bagh, B., McKinty, A.M., Lough, A.J., et al., Dalton Trans., 2015, vol. 44, no. 6, p. 2712.
Rayati, S. and Salehi, F., J. Iran. Chem. Soc., 2015, vol. 12, no. 2, p. 309.
Haque, M.R., Ghosh, S., Hogarth, G., et al., Inorg. Chim. Acta, 2015, vol. 434, p. 150.
Szatkowski, L. and Hall, M.B., Inorg. Chem., 2015, vol. 54, no. 13, p. 6380.
Bagherzadeh, M., Amini, M., Parastar, H., et al., Inorg. Chem. Commun., 2012, vol. 20, no. 1, p. 86.
Dupe, A., Hossain, M.K., Schachner, J.A., et al., Eur. J. Inorg. Chem., 2015, no. 21, p. 3572.
Oliveira, T.S.M., Gomes, A.C., Lopes, A.D., et al., Dalton Trans., 2015, vol. 44, no. 31, p. 14139.
Mirzaee, M., Bahramian, B., and Amoli, A., Appl. Organomet. Chem., 2015, vol. 29, no. 9, p. 593.
Jones, M.M., J. Am. Chem. Soc., 1959, vol. 81, no. 6, p. 3188.
Sheldrick, G.M., SADABS, Göttingen: Univ. of Göttingen, 1996.
Sheldrick, G.M., SHELX-97, Program for Crystal Structure Solution and Refinement, Göttingen: Univ. of Göttingen, 1997.
Sheldrick, G.M., SHELXTL, Version 5, Siemens, Madison: Industrial Automation Inc., 1995.
Geary, W.J., Coord. Chem. Rev., 1971, vol. 7, no. 1, p. 81.
Gupta, S., Barik, A.K., Pal, S., et al., Polyhedron, 2007, vol. 26, no. 1, p. 133.
Ngan, N.K., Lo, K.M., and Wong, C.S.R., Polyhedron, 2012, vol. 33, no. 1, p. 235.
Bagherzadeh, M., Latifi, R., Tahsini, L., et al., Polyhedron, 2009, vol. 28, no. 12, p. 2517.
Nunes, C.D., Valente, A.A., Pillinger, M., et al., Chem. Eur. J., 2003, vol. 9, no. 18, p. 4380.
Kuhn, F.E., Groarke, M., Bencze, E., et al., Chem. Eur. J., 2002, vol. 8, no. 10, p. 2370.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Zhang, W.G., Liang, J.H. Synthesis, characterization, and crystal structures of dioxomolybdenum(VI) complexes with O,N,N type tridentate hydrazone ligands as catalyst for oxidation of olefins. Russ J Coord Chem 43, 411–419 (2017). https://doi.org/10.1134/S1070328417060100
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328417060100