Abstract
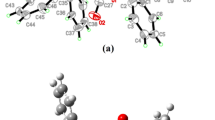
Mild solvothermal synthesis, structures, thermal and magnetic properties of coordination complexes [Ln(PDA)2(PDAH2)] · (DMAH2)2(DMAH0.5)2(I–IV) (PDA = pyrdine-2,6-dicarboxylate anion, DMAH = dimethylamine, Ln = Ce, Nd, Sm, and Ho) are described. The DMAH molecules in I–IV, generated in situ from hydrolysis of N,N-dimethylformamide, are responsible to assemble three dimensional coordination polymers through N–H···O and C–H···O hydrogen bonds. Distorted tricapped trigonal prismatic LnO6N3 geometry having 14 triangular faces is attributed to mean deviation of dihedral angles while nitrogen shows fairly triangular faces having dihedral angle close to 60°C (CIF files CCDC nos. 872065 (I), 872070 (II), 872069 (III), and 872066 (IV)). Curie–Weiss law and the overall magnetic behavior are typical for the presence of antiferromagnetic exchange coupling interactions between lanthanide. Thermal decomposition analyses reveal removal of ammonia and resultant complexes showthermal stability. Complexes have been further characterized by using elemental analyzer and FT-IR spectroscopy.
Similar content being viewed by others
References
Eddaoudi, M., Kim, J., Rosi, N., et al., Science, 2002, vol. 295, p. 469.
Allendorf, M.D., Bauer, C.A., Bhakta, R.K., et al., Chem. Soc. Rev., 2009, vol. 38, p. 1330.
Huang, Y.G., Jiang, F.L., and Hong, M.C., Coord. Chem. Rev., 2009, vol. 253, p. 2814.
Munakata, M., Ning, G.L., Kuroda-Sowa, T., et al., Inorg. Chem., 1998, vol. 37, p. 5651.
Losier, P. and Zaworotko, M.J., Angew. Chem. Int. Ed., 1996, vol. 35, p. 2779.
Wen, L.L., Wang, F.-M., Leng, X.-K., et al., J. Inorg. Organomet. Polym. Mater., 2010, vol. 20, p. 313.
Gable, R.W., Hoskins, B.F., and Robson, R., J. Chem. Soc., Chem. Commun., 1990, p. 1677.
Desiraju, G.R., Angew. Chem. Int. Ed., 1995, vol. 34, p. 2311.
Schauer, C.L., Matwey, E., Fowler, F.W., et al., J. Am. Chem. Soc., 1997, vol. 119, p. 10245.
Crystal Design: Structure and Function, Desiraju, G.R., Ed., New York: Wiley, 2003, p. 4.
Khan, I.U., Sharif, S., and Sahin, O., J. Coord. Chem., 2013, vol. 66, p. 3113.
Sharif, S., Khan, I.U., Sahin, O., et al., J. Inorg. Organomet., 2012, vol. 22, p. 1165.
Macgillivray, L.R., Metal-Organic Frameworks Design and Application, Hoboken (NJ, USA): Wiley, 2010, p. 13.
Feng, M.-L., Xie, Z.-L., and Huang, X.-Y., Inorg. Chem., 2009, vol. 48, p. 3904.
Feng, M.L., Kong, D.N., Xie, Z.L., et al., Angew. Chem., Int. Ed., 2008, vol. 47, p. 8623.
Zhou, Y.-X., Cheng, G.-X., Wuc, B.-L., et al., Acta Crystallogr., Sect. E: Struct. Rep. Online, 2007, vol. 63, p. m2285.
Xu, Y.L., Han, Z., Lin, Z., et al., Eur. J. Inorg. Chem., 2004, p. 4457.
Sheldrick, G.M., Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, vol. 64, p. 112.
Betteridge, P.W., Carruthers, J.R., Cooper, R.I., et al., J. Appl. Cryst., 2003, vol. 36, p. 1487.
Farrugia, L.J., J. Appl. Cryst., 1999, vol. 32, p. 837.
Macrae, C.F., Bruno, I.J., Chisholm, J.A., et al., J. Appl. Cryst., 2008, vol. 41, p. 466.
Spek, A.L., J. Appl. Crystallogr., 2003, vol. 26, p. 1.
Crystal and Molecular Structures Program for Mac and Windows, CrystalMaker® software, www.crystalmaker.com
OriginPro 8 SRO, v. 8.0724 (B724), Northampton (MA, USA), www.originLab.com
Blundell, S. and Thouless, D., Magnetism in Condensed Matter, Oxford: Oxford Univ., 2001.
Want, B., Ahmad, F., and Kotru, P.N., J. Alloys Compd., 2008, vol. 448, p. L5.
Prasad, T.K. and Rajasekharan, M.V., Cryst. Growth Des., 2008, vol. 8, p. 1346.
Wang, H.S., Zhao, B., Zhai, B., et al., Cryst Growth Des., 2007, vol. 7 p. 1851.
Gao, H.L., Zhao, B., Zhao, X.Q., et al., Inorg. Chem., 2008, vol. 47, p. 11057.
Karroker, D.G., J. Chem. Ed., 1970, vol. 47, p. 424.
Keller, R.N., J. Chem. Ed., 1951, p. 312.
Rare Earth Coordination Chemistry Fundamentals and Applications, Huang, C., Ed., Wiley (Asia), 2010, p. 47.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Sharif, S., Khan, B., Şahin, O. et al. Lanthanide complexes with pyridine-2,6-dicarboxylic acid: synthesis, crystal structure, thermal and magnetic properties of [LnPDA)2(PDAH2)] · (DMAH2)2(DMAH0.5)2 . Russ J Coord Chem 42, 56–65 (2016). https://doi.org/10.1134/S1070328416010048
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328416010048