Abstract
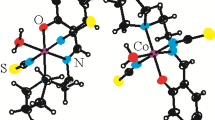
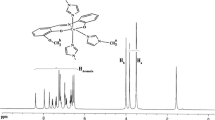
The synthesis, characterization, spectroscopic and electrochemical properties of trans-[CoIII(L1)(Py)2]ClO4 (I) and trans-[CoIII(L2)(Py)2]ClO4 (II) complexes, where H2L1 = N,N′-bis(5-chloro-2-hydroxybenzylidene)-1,3-propylenediamine and H2L2 = N,N′-bis(5-bromo-2-hydroxybenzylidene)-1,3-propylenediamine, have been investigated. Both complexes have been characterized by elemental analysis, FT-IR, UV-Vis, and 1H NMR spectroscopy. The crystal structure of I has been determined by X-ray diffraction. The coordination geometry around cobalt(III) ion is best described as a distorted octahedron. The electrochemical studies of these complexes revealed that the first reduction process corresponding to Co(III/II) is electrochemically irreversible accompanied by dissociation of the axial Co-N(Py) bonds. The in vitro antimicrobial activity of the Schiff bse ligands and their corrsponding complexes have been tested against human pathogenic bacterias such as Staphylococcus aureus, Bacillus subtilis, Pseudomonas aeruginosa, and Escherichia coli. The cobalt(III) complexes showed lower antimicrobial activity than the free Schiff base ligands.
Similar content being viewed by others
References
Cimernman, Z., Galic, N., and Bosner, B., Anal. Chim. Acta, 1997, vol. 343, p. 145.
Leeb, M., Nature, 2004, vol. 431, p. 892.
Patil, S.A., Naik, V.H., Kulkarni, A.D., and Badami, P.S., J. Mol. Struct., 2011, vol. 985, p. 330.
Yamada, S., Coord. Chem. Rev., 1999, vols. 190–192, p. 537.
Bianchini, C. and Zoeliner, R.W., Adv. Inorg. Chem., 1997, vol. 44, p. 263.
Zhu, Y. and Li, W.-H., Transition Met. Chem., 2010, vol. 35, p. 745.
Amirnasr, M., Shenk, K.J., Gorji, A., and Vafazadeh, R., Polyhedron, 2001, vol. 20, p. 695.
Zhang, Y.L., Ruan, W.J., Zhao, X.J., et al., Polyhedron, 2003, vol. 22, p. 1535.
Salehi, M., Dutkiewicz, G., and Kubicki, M., Acta Crystallogr., E, 2010, vol. 66, p. 1590.
Schenk, K.J., Meghdadi, S., Amirnasr, M., et al., Polyhedron, 2007, vol. 26, p. 5448.
Elerman, Y., Elmali, Y., Kabak, M., and Svoboda, I., Acta Crystallogr., C, 1998, vol. 54, p. 1701.
Agilent Technologies, CRYSALIS PRO, Version 1.171.33.36d, Oxford Diffraction Ltd., 2010.
Altomare, A., Cascarano, G., Giacovazzo, C., and Gualardi, A., J. Appl. Crystallogr., 1993, vol. 26, p. 343.
Sheldrick, G.M., Acta Crystallogr., A, 2008, vol. 64, p. 112.
Bauer, A.W., Kirby, W.M., Sheris, J.C., and Turck, M., Am. J. Clin. Pathol., 1966, vol. 45, p. 493.
Goldman, E. and Green, L.H., Practical Handbook of Microbiology, New York: CRC Press, Taylor & Francis Group, 2009, p. 37.
European Committee for Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID), Clin. Microbiol. Infect., 2000, vol. 6, p. 509.
Anthonysamy, A. and Balasubramanian, S., Inorg. Chem. Commun., 2005, vol. 8, p. 908.
Garg, B.S. and Kumar, D.N., Spectrochim. Acta, A, 2003, vol. 59, p. 229.
Amirnasr, M., Vafazadeh, R., and Mahmoudkhani, A., Can. J. Chem., 2002, vol. 80, p. 1196.
Nakamoto, K., Infrared Spectra of Inorganic Compounds, New York: Wiley, 1970.
Zamian, J.R. and Dockal, E.R., Transition Met. Chem., 1996, vol. 21, p. 370.
Felicio, R.C., da Silva, G.A., Ceridorio, L.F., and Dockal, E.R., Synth. React. Inorg. Met. Org. Chem., 1999, vol. 29, p. 171.
Signorini, O., Dockal, E.R., Castellano, G., and Oliva, G., Polyhedron, 1996, vol. 15, p. 245.
Zhang, Y.-L., Ruan, W.-J., Zhao, X.-J., et al., Polyhedron, 2003, vol. 22, p. 1535.
Bottche, A., Takeuchi, T., Hardcastle, K.I., et al., Inorg. Chem., 1997, vol. 36, p. 2498.
Kruger, G.J. and Reynhardt, E.C., Acta Crystallogr., B, 1978, vol. 34, p. 915.
Shi, X.-H., You, X.-Z., Li, C., et al., Acta Crystallogr., C, 1995, vol. 51, p. 206.
Nejo, A.A., Kolawole, G.A., and Nejo, A.O., J. Coord. Chem., 2010, vol. 63, p. 4398.
Performance Standards for Antimicrobial Susceptibility Testing, M100-S19, Wayne (PA, USA): Clinical and Laboratory Standards Institute, 2009.
Mishra, A., Kaushik, N.K., Verma, A.K., and Gupta, R., Eur. J. Med. Chem., 2008, vol. 43, p. 2189.
Jorgensen, J.H. and Ferraro, M.J., Clin. Infect. Dis., 2009, vol. 49, p. 1749.
ÖZdemir, Ü.Ö., Arslan, F., and Hamurcu, F., Inorg. Chim. Acta, 2009, vol. 362, p. 2613.
Chohan, Z.H., Hassan, M.U., Khan, K.M., and Supuran, C.T., J. Enzym. Inhib. Med. Chem., 2005, vol. 20, p. 183.
Zanatta, N., Alues, S.H., Coelho, H.S., et al., Bioorg. Med. Chem., 2007, vol. 15, p. 1947.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Salehi, M., Kubicki, M., Dutkiewicz, G. et al. Synthesis, characterization, electrochemical studies, and antibacterial activities of cobalt(III) complexes with Salpn-Tipe Schiff base ligands. Crystal structure of trans-[CoIII(L1)(Py)2]ClO4 . Russ J Coord Chem 39, 716–722 (2013). https://doi.org/10.1134/S1070328413100084
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328413100084