Abstract
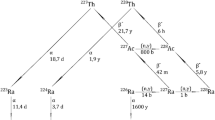
A target containing 3.16 mg of 226RaCO3 was irradiated in the neutron trap of the SM reactor for 25 effective days at a thermal neutron flux of 1.5 × 1015 cm−2 s−1. After the irradiation and storage for 17 days, the irradiated material was dissolved and the Ra activation products were chemically isolated. The yields of 229Th, 230Th, 227Ac, and 228Th were determined by α- and γ-ray spectroscopy and by mass spectrometry. Formation of significant amounts of 228Ra, not predicted by the calculations, was revealed. It was suggested that the short-lived isotope 227Ra has a high neutron capture cross section [σeff(227Ra) ≈ 1.5 × 103 b].
Similar content being viewed by others
References
Sgouros, G., Ballangrud, A.M., Jurcic, J.G., et al., J. Nucl. Med., 1999, vol. 40, no. 11, pp. 1935–1946.
Chemotherapy and Monoclonal Antibody Therapy in Treating Patients with Advanced Myeloid Cancer, http://www.clinicaltrials.gov/ct2/show/NCT00014495.
Protein Design Labs Licenses Development Rights to SMART M195 Antibody to Actinium Pharmaceuticals, Actinium Pharmaceuticals Inc. Press Release from March 18, 2003.
Targeted Atomic Nano-Generators (Actinium-225-Labeled Humanized Anti-CD33 Monoclonal Antibody HuM195) in Patients with Advanced Myeloid Malignancies, http://www.clinicaltrials.gov/ct2/show/study/NCT00672165.
Nilsson, S., Larsen, R.H., Fossa, S., et al., Clin. Cancer Res., 2005, vol. 11, no. 12, pp. 4451–4459.
A Phase III Study of Alpharadin (Radium-223) in Patients with Symptomatic Hormone Refractory Prostate Cancer with Skeletal Metastases (ALSYMPCA), http://www.clinicaltrials.gov/ct2/show/NCT00699751.
A Study of Alpharadin® with Docetaxel in Patients with Bone Metastasis from Castration-Resistant Prostate Cancer (CRPC), http://www.clinicaltrials.gov/ct2/show/NCT01106352.
Wesley, J.N., McGee, E.C., Garmestani, K., et al., Nucl. Med. Biol., 2004, vol. 31, pp. 357–364.
Milenic, D.E., Garmestani, K., Brady, E.D., et al., Clin. Cancer Res., 2008, vol. 14, pp. 5108–5115.
Horak, E., Hartmann, F., Garmestani, K., et al., J. Nucl. Med., 1997, vol. 38, no. 12, pp. 1944–1950.
Dahle, J., Borrebaek, J., Melhus, K.B., et al., Nucl. Med. Biol., 2006, vol. 33, pp. 271–279.
Forsberg, C.W. and Lewis, L.C., Uses for Uranium-233: What Should Be Kept for Future Needs?, ORNL, 1999, p. 19.
Vakhetov, F.Z. and Toporov, Yu.G., Sb. Tr. NIIAR, 2004, vol. 1, pp. 29–38.
Karalova, Z.K., Ivanov, R.N., Myasoedov, B.F., et al., At. Energ., 1972, vol. 32, no. 2, pp. 119–121.
Sarig, S. and Kahana, F., Thermochim. Acta, 1976, vol. 14, no. 3, pp. 263–268.
MCNP-A General Monte Carlo N-Particle Transport Code, Version 4C, Briesmeister, J.F., Ed., Rep. LA-13709-M, 2000.
Atcher, R.W., Hines, J.J., and Fiedman, A.M., J. Radioanal. Nucl. Chem. Lett., 1987, vol. 117, no. 3, p. 155.
Rosman, K.J.R. and Taylor, P.D.P., Pure Appl. Chem., 1998, vol. 70, no. 1, pp. 217–236.
RSICC Newsletters. Radiation Safety Information Computational Center, Oak Ridge National Laboratory, January 2006, no. 491, p. 1.
Romanov, E.G., A Tool to Calculate Nuclear Transmutations in a Neutron Flux. Research Center for Atomic Reactors: IAEA 1404 Code, 2003.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © R.A. Kuznetsov, P.S. Butkalyuk, V.A. Tarasov, A.Yu. Baranov, I.L. Butkalyuk, E.G. Romanov, V.N. Kupriyanov, E.V. Kazakova, 2012, published in Radiokhimiya, 2012, Vol. 54, No. 4, pp. 352–356.
Rights and permissions
About this article
Cite this article
Kuznetsov, R.A., Butkalyuk, P.S., Tarasov, V.A. et al. Yields of activation products in 226Ra irradiation in the high-flux SM reactor. Radiochemistry 54, 383–387 (2012). https://doi.org/10.1134/S1066362212040121
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1066362212040121