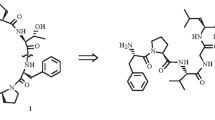
Abstract
Boc-Δ3Pro-Pro-Ile-OBzl and Boc-Pro-Δ3Pro-Ile-OBzl were synthesized. Tritium-labeled Pro-Pro-Ile was prepared by their hydrogenation in a tritium atmosphere in the presence of a catalyst. In the reduction of Boc-Δ3Pro-Pro-Ile-OBzl and Boc-Pro-Δ3Pro-Ile-OBzl, elimination of the Bzl protective group occurs faster than hydrogenation of the 3,4-dehydroproline residue (benzene and dioxane were used as solvents). The resulting Boc-Δ3Pro-Pro-Ile-OH and Boc-Pro-Δ3Pro-Ile-OH are poorly soluble in aprotic solvents, which accounts for the low yield of the desired tritium-labeled peptides.
Similar content being viewed by others
References
Makarenko, E.Yu., Andreeva, L.A., Borovik, A.S., and Mart’yanov, A.A., Ross. Fiziol. Zh. im. I.M. Sechenova, 2003, vol. 89, no. 8, pp. 943–950.
Shevchenko, V.P., Nagaev, I.Yu., and Myasoedov, N.F., Mechennye tritiem lipofil’nye soedineniya (Tritium-Labeled Lipophilic Compounds), Moscow: Nauka, 2003.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.P. Shevchenko, I.Yu. Nagaev, L.A. Andreeva, N.F. Myasoedov, 2010, published in Radiokhimiya, 2010, Vol. 52, No. 3, pp. 277–280.
Rights and permissions
About this article
Cite this article
Shevchenko, V.P., Nagaev, I.Y., Andreeva, L.A. et al. Synthesis of Pro-Pro-Ile, a corticoliberin fragment, labeled with tritium in proline unit. Radiochemistry 52, 325–329 (2010). https://doi.org/10.1134/S1066362210030185
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1066362210030185