Abstract
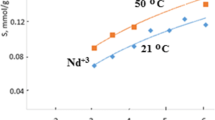
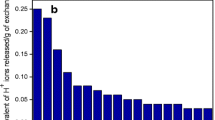
The kinetics of extraction of uranyl nitrate from aqueous salt solutions with a composite material based on a polymeric support and trialkylmethylammonium nitrate at T = 293.15–333.15 K in the presence of 1.0–4.0 M NaNO3 in the aqueous phase was studied. The rate-determining step of the process is diffusion through the phase boundary (film kinetics). The mass transfer coefficients were determined, and from their temperature dependence the apparemt activation energy (E a) was estimated. The mass transfer coefficients increase with an increase in temperature and in the NaNO3 concentration in the aqueous phase, whereas E a is virtually independent of the NaNO3 concentration and equal to 26.4 ± 1.2 kJ mol−1.
Similar content being viewed by others
References
Pyartman, A.K., Keskinov, V.A., Lishchuk, V.V., et al., Zh. Prikl. Khim., 2006, vol. 79, no. 8, pp. 1822–1826.
Pyartman, A.K., Keskinov, V.A., Mikhailenko, M.A., et al., Radiokhimiya, 2004, vol. 46, no. 3, pp. 240–242.
Pyartman, A.K., Kopyrin, A.A., Berinskii, A.E., and Keskinov, V.A., Radiokhimiya, 2000, vol. 42, no. 2, pp. 128–130.
Přibil, R., Analytical Applications of EDTA and Related Compounds, Oxford: Pergamon, 1972.
Pyartman, A.K., Keskinov, V.A., and Lishchuk, V.V., Radiokhimiya, 2008, vol. 50, no. 2, pp. 137–141.
Pyartman, A.K., Lishchuk, V.V., and Keskinov, V.A., Radiokhimiya, 2008, vol. 50, no. 2, pp. 142–143.
Osnovy zhidkostnoi ekstraktsii (Principles of Liquid Extraction), Yagodin, G.A., Ed., Moscow: Khimiya, 1981.
Zelikman, A.I., Vol’dman, G.M., and Belyavskaya, L.V., Teoriya gidrometallurgicheskikh protsessov (Theory of Hydrometallurgical Processes), Moscow: Metallurgiya, 1975.
Treybal, R.E., Liquid Extraction, New York: McGraw-Hill, 1963.
Author information
Authors and Affiliations
Additional information
Original Russian Text © V.A. Keskinov, A.K. Pyartman, 2008, published in Radiokhimiya, 2008, Vol. 50, No. 5, pp. 414–415.
Rights and permissions
About this article
Cite this article
Keskinov, V.A., Pyartman, A.K. Kinetics of extraction of uranyl nitrate from aqueous salt solutions with a composite material based on a polymeric support and trialkylmethylammonium nitrate at various temperatures. Radiochemistry 50, 479–481 (2008). https://doi.org/10.1134/S1066362208050081
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1066362208050081