Abstract
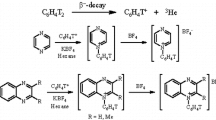
Ion-molecule reactions of free phenyl cations with six-membered nitrogen-containing heterocyclic compounds: α-, β-, and γ-picolines, were studied. Phenyl cations were generated by tritium β-decay in double-labeled benzene. The mechanism of competing reactions of electrophilic addition to the nitrogen heteroatom in the ring and electrophilic substitution of the C-H bond in the pyridine ring was examined. The effect of methyl substituent in the pyridine molecule on the reaction pathway was evaluated. A one-step procedure for nuclear-chemical synthesis of tritium-labeled N-phenylpicolinium salts and phenyl-substituted picolines was developed.
Similar content being viewed by others
References
Saljoughian, M., Synthesis, 2002, vol. 13, pp. 1781–1801.
Shevchenko, V.P., Nagaev, I.Yu., and Myasoedov, N.F., Mechennye tritiem lipofil’nye soedineniya (Tritium-Labeled Lipophilic Compounds), Moscow: Nauka, 2003.
Ivanskii, V.I., Khimiya geterotsiklicheskikh soedinenii (Chemistry of Heterocyclic Compounds), Moscow: Vysshaya Shkola, 1978.
Bettica, P., Masino, M., Cucinotta, E., et al., Eur. J. Clin. Chem. Clin. Biochem., 1997, vol. 35, pp. 63–68.
Rao, K.N., Redda, K.K., and Onayemi, F.Y., J. Heterocyclic Chem., 1995, vol. 32, pp. 307–315.
Goldmam, R.C., Zakula, D., Capobianco, J.O., et al., Antimicrob. Agents Chemother., 1996, vol. 40, pp. 1044–1047.
Whiteley, C.G. and Ngwenya, D.S., Biochem. Biophys. Res. Commun., 1995, vol. 211, pp. 1083–1090.
Alaiz, M., Zamora, R., and Hidalgo, F.J., J. Agric. Food Chem., 1996, vol. 44, pp. 686–691.
Igarashi, K., Matsubara, K., Kasuya, F., et al., Neurosci. Lett., 1996, vol. 214, pp. 183–186.
Marowska, J., Kobylinska, M., Lukaszkiewicz, J., et al., Bone, 1996, vol. 19, pp. 669–677.
Eyless, D.W., Avent, K.M., Stedman, T.J., and Pond, S.M., Life Sci., 1997, vol. 60, pp. 529–534.
Pozharskii, A.F., Soldatenkov, A.T., and Katritzky, A.R., Heterocycles in Life and Society, New York: Wiley, 1997.
Gilchrist, T.L., Heterocyclic Chemistry, Harlow: Longman, 1992, 2nd ed.
Petucci, C., Nyman, M., Guler, L., and Kenttamaa, H., J. Am. Chem. Soc., 2002, vol. 124, pp. 4108–4115.
Ayers, J.T., Dwoskin, L.P., Deaciuc, A.G., et al., Bioorg. Med. Chem. Lett., 2002, vol. 12, no. 21, pp. 3067–3071.
Freerich, M., Cristino, A., Choil, Y.H., et al., Planta Med., 2004, vol. 1, pp. 72–76.
Foster, S.B., Tang, H., Miller, K.E., and Dryhurst, G., Neurotox. Res., 2005, vol. 7, no. 4, pp. 251–263.
Dorofenko, G.N., Sadekova, E.I., and Kuznetsov, E.V., Preparativnaya khimiya pirilievykh solei (Preparative Chemistry of Pyrylium Salts), Rostov-on-Don: Rostov. Univ., 1972.
Coe, B.J., Horris, J.A., Asselbeighs, J., and Kloys, K., Adv. Funct. Mater., 2002, vol. 12, no. 2, pp. 110–116.
Suzuki, A., J. Organomet. Chem., 1999, vol. 576, p. 147.
Shchepina, N.E., Nefedov, V.D., Toropova, M.A., et al., Radiokhimiya, 1999, vol. 41, no. 6, pp. 523–525.
Shchepina, N.E., Nefedov, V.D., Toropova, M.A., et al., Tetrahedron Lett., 2000, vol. 41, no. 1, pp. 25–29.
Katritzky, A.R. and Taylor, R., Adv. Heterocyclic Chem., 1990, vol. 47, p. 1.
Author information
Authors and Affiliations
Additional information
Original Russian Text © N.E. Shchepina, V.V. Avrorin, G.A. Badun, V.M. Fedoseev, S.E. Ukhanov, S.B. Lewis, 2007, published in Radiokhimiya, 2007, Vol. 49, No. 6, pp. 551–553.
Rights and permissions
About this article
Cite this article
Shchepina, N.E., Avrorin, V.V., Badun, G.A. et al. Nuclear-chemical synthesis of tritium-labeled phenyl-substituted picoline derivatives. Radiochemistry 49, 630–632 (2007). https://doi.org/10.1134/S1066362207060161
Received:
Issue Date:
DOI: https://doi.org/10.1134/S1066362207060161