Abstract
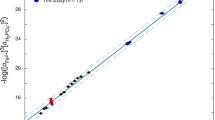
The solubility of UO2HPO4 in normal seawater of 35‰ salinity was studied. The solubility of U(VI) phosphates in seawater increases nonlinearly with decreasing acidity (by two orders of magnitude per pH unit) due to formation of strong hydroxo, carbonate and, probably, phosphate complexes. At pH > 7.5, UO2HPO4 dissolves congruently. With decreasing pH to 6.9–7.2, UO2HPO4 is converted to UO2(H2PO4)2. The solubility of U(VI) phosphates in seawater considerably exceeds the natural uranium concentrations in seawater and pore solutions of the bottom sediments. Therefore, these compounds cannot form intrinsic minerals in the oceanic bottom sediments. The possibility of accumulation of U(VI) in sea phosphorites due to its chemisorption in the form of surface uranyl phosphate complexes is discussed.
Similar content being viewed by others
References
Savenko, V.S., Dokl. Akad. Nauk SSSR, 1978, vol. 243, no. 5, pp. 1302–1305.
Golubev, S.V., Savenko, V.S., and Pokrovskii, O.S., Vest. Mosk. Univ., Ser. 4: Geol., 2000, no. 2, pp. 71–76.
Savenko, V.S., Fiziko-khimicheskii analiz protsessov formirovaniya zhelezomargantsevykh konkretsii v okeane (Physicochemical Analysis of Formation of Iron-Manganese Nodules in Ocean), Moscow: GEOS, 2004.
Baturin, G.N., Uran v sovremennom morskom osadkoobrazovanii (Uranium in the Modern Sea Sedimentation), Moscow: Atomizdat, 1975.
Baturin, G.N., Fosfority na dne okeanov (Phosphorites at Ocean Bottom), Moscow: Nauka, 1978.
Author information
Authors and Affiliations
Additional information
Original Russian Text © A.V. Savenko, 2006, published in Radiokhimiya, 2006, Vol. 48, No. 2, pp. 150–152.
Rights and permissions
About this article
Cite this article
Savenko, A.V. Solubility of UO2HPO4 in seawater. Radiochemistry 48, 167–169 (2006). https://doi.org/10.1134/S1066362206020123
Received:
Issue Date:
DOI: https://doi.org/10.1134/S1066362206020123