Abstract
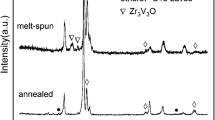
The process of hydrogen decrepitation of the (Nd, Pr, Dy)(Fe, Co)2.6 alloy in wide temperature and pressure ranges has been studied. The choices of the chemical composition of the (Nd, Pr, Dy)(Fe, Co)2.6 alloy and the method of production of the initial alloy based on out-of-furnace calcium–thermal reduction are substantiated. The introduction of dysprosium into the alloy composition contributes to an increase in the coercive force. Additional alloying with cobalt is related to the need to increase the Curie temperature for this class of magnetic materials. Investigations have been carried out on two groups of alloys. The first group of alloys is the initial one. The second group is loaded into a steel reactor and subjected to isothermal annealing (heat treatment) in vacuum at 833 K for 1 h with subsequent supply of argon and quenching of the samples by immersing the reactor (containing the samples) in water. It is established by scanning electron microscopy that heat treatment leads to the formation of microcracks and increases the relief on the sample surface. The initial and annealed samples have been subjected to hydrogenation at different temperatures and pressures, which facilitates their transition to the powder state. Hydrogenation has been performed using high-purity hydrogen obtained by its desorption from a LaNi5 alloy. The hydrogenation process has been performed at temperatures from 298 to 473 K and pressures from 100 to 200 kPa. It is established that the highest degrees of hydrogenation are obtained at low pressures and temperatures. It is also found that the induction period for the annealed alloy is 432 s, in contrast to 3200 s for the alloy without heat treatment.





Similar content being viewed by others
REFERENCES
A. Walton, Y. Han, N. A. Rowson, et al., J. Cleaner Prod. 104, 236 (2015).
J. Luo, P. Rango, D. Fruchart, J. N. Mei, and L. Zhou, J. Alloys Compd. 509, 4252 (2011).
X. H. Wang, G. C. Yin, C. Y. Cu, et al., J. Alloys Compd. 726, 846 (2017).
J. Jin, M. Yan, Y. Liu, et al., Acta Mater. 169, 248 (2019).
Y. M. Tang, Y. He, Y. Huang, et al., J. Magn. Magn. Mater. 451, 515 (2018).
N. B. Kol’chugina, Doctoral Dissertation in Engineering (Moscow, 2018).
M. Zakotnik, A. J. Williams, A. G. Martinek, and I. R. Harris, J. Alloys Compd. 450, L1 (2008).
X. Yin, M. Yue, Q. Lu, et al., Engineering 6, 165 (2020).
H. Nakamura, K. Hirota, M. Shimao, et al., IEEE Trans. Magn. 41, 3844 (2005).
A. Kianvash, R. S. Mottram, and I. R. Harris, J. Alloys Compd. 287, 206 (1999).
T.-H. Kim, S.-R. Lee, H.-J. Kim, et al., J. Appl. Phys. 115, 17A763 (2014).
E. Yu. Kartashov, Candidate’s Dissertation in Engineering (Seversk, 2006).
L. A. Stashkova, V. S. Gaviko, N. V. Mushnikov, and P. B. Terent’ev, Phys. Met. Metallogr. 114 (12), 985 (2013). https://doi.org/10.1134/S0031918X13120077
A. S. Buinovskii et al., Fluoride Technology for the Production of Magnetic Materials Based on Rare Earth Elements for Nuclear Power, Part I: Extra-Furnace Fluoride Technology of Rare Earth Alloys (TUSUR, Tomsk, 2012) [in Russian].
H. Zhang, Z. Wang, F. Liu, et al., Russ. J. Electrochem. 51 (3), 258 (2015). https://doi.org/10.1134/S102319351503012X
V. N. Verbetskii, Doctoral Dissertation in Engineering (Moscow, 1998).
N. A. Kulabukhova, G. M. Poletaev, M. D. Starostenkov, et al., Izv. Vyssh. Uchebn.Zaved., Fiz., No. 12, 86 (2011).
I. I. Bulyk, R. V. Denys, V. V. Panasyuk, et al., Mater. Sci. 37, 544 (2001).
D. V. Shchur, S. Yu. Zaginaichenko, A. Veziroglu, et al., Al’tern. Energ. Ekol., Nos. 13–15, 62 (2019).
M. H. Mintz and J. Bloch, J. Chem. Phys. 78, 6569 (1983).
Funding
This study was supported by the Ministry of Science and Higher Education of the Russian Federation within the framework of state contract no. FEMN-2020-0004.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by A. Sin’kov
Rights and permissions
About this article
Cite this article
Grachev, E.K., Buinovsky, A., Muslimova, A. et al. Investigation of the Process of Hydrogen Dispersion of the (Nd, Pr, Dy)(Fe, Co)2.6 Alloy in Specified Temperature and Pressure Ranges. Tech. Phys. 67, 743–748 (2022). https://doi.org/10.1134/S1063784222100103
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1063784222100103