Abstract
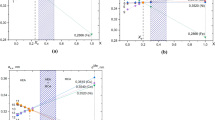
An original technique of computer modeling of substitutional solid solutions has been applied to Al2O3-Cr2O3, Al2O3-Fe2O3, and Fe2O3-Cr2O3 binary systems. The parameters of semiempirical interatomic potentials were optimized using the experimentally studied structural, elastic, and thermodynamic properties of pure components. Among point defects, the most energetically favorable ones for all three oxides are Schottky vacancy quintets. To model (M 1x M 21 − x )2O3 solid solutions, 4 × 4 × 1 disordered supercells with M 1: M 2 cation ratios of 1: 5, 1: 2, 1: 1, 2: 1, and 5: 1 have been constructed in the cation sublattice containing 192 atoms. The mixing enthalpy and volume, interaction parameters, bulk moduli, and vibrational entropy were found by minimizing the interatomic interaction energy in supercells with the symmetry P1. Calculations of the Gibbs energy made it possible to estimate the fields of stability of the Al2O3-Cr2O3 and Al2O3-Fe2O3 solid solutions; these estimates were compared with the experimental data. Histograms of M-M, M-O, and O-O interatomic distances were constructed and the local structure was analyzed for the Al1.0Cr1.0O3, Al1.0Fe1.0O3, and Fe1.0Cr1.0O3 compositions.
Similar content being viewed by others
References
V. S. Urusov, T. G. Petrova, and N. N. Eremin, Dokl. Akad. Nauk 387, 191 (2002).
V. S. Urusov, T. G. Petrova, and N. N. Eremin, Dokl. Akad. Nauk 392, 1 (2003).
N. N. Eremin, R. Deyanov, and V. S. Urusov, in Proceedings of the International Conference “Spectroscopy and Crystal Chemistry of Minerals,” Yekaterinburg, Russia, 2007, p. 40.
K. T. Jacob, J. Electrochem. Soc. 125, 175 (1978).
N. D. Chatterjee, et al., Am. Mineral. 67, 725 (1982).
T. Grygar et al., Ceramics, Silikaty 47(1), 32 (2003).
S. Music et al., J. Mater. Sci. 31, 4067 (1996).
Y. Murakami, A. Sawata, and Y. Tsuru, J. Mater. Sci. 34, 951 (1999).
J. Majzlan, A. Navrotsky, and B. J. Evans, Phys. Chem. Miner. 29, 515 (2002).
A. Feenstra, S. Sämann, and B. Wunder, J. Petrol. 46(9), 1881 (2005).
J. D. Gale and A. L. Rohl, Mol. Simul. 29(5), 291 (2003).
N. N. Eremin, R. Z. Deyanov, and V. S. Urusov, Fiz. Khim. Stekla 34(1), 11 (2008).
V. S. Urusov, N. N. Eremin, T. G. Petrova, and R. A. Talis, in Proceedings of the IV National Conference on Crystal Chemistry, Chernogolovka, Russia, 2006, p. 238.
C. R. A. Catlow, J. D. Gale, and R. W. Grimes, J. Solid State Chem. 106, 13 (1993).
J. D. Gale, C. R. A. Catlow, and W. C. Mackrodt, Mod. Simul. Mater. Sci. Eng. 1, 73 (1992).
R. A. Robie and B. S. Hemingway, U. S. Geological Survey Bulletin no. 2131 (U. S. Government Printing Office, Washington, 1995).
T. Goto, S. Yamamoto, I. Ohno, and O. L. Anderson, J. Geophys. Res. 94, 7588 (1989).
N. Ishimata, T. Miyata, J. Minato, et al., Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem. 36, 228 (1980).
L. Finger and R. M. Hazen, J. Appl. Phys. 51, 5362 (1980).
M. Iu. Antipin, V. G. Tsirelson, M. P. Flugge, et al., Dokl. Akad. Nauk SSSR 281, 845 (1985) [Sov. Phys. Dokl. 30, 306 (1985)].
G. J. Dienis, D. O. Welch, C. R. Fisher, et al., Phys. Rev. B: Solid State 11, 3060 (1975).
C. R. A. Catlow, R. James, W. C. Mackrodt, and R. F. Stewart, Phys. Rev. B: Condens. Matter 25, 1006 (1992).
P. W. M. Jacobs and E. A. Kotomin, J. Solid State Chem. 106, 27 (1993).
S. K. Mohapatra and F. A. Kroger, J. Am. Ceram. Soc. 61, 106 (1978).
V. S. Urusov, V. L. Tauson, and V. V. Akimov, Geochemistry of Solids (GEOS, Moscow, 1997) [in Russian].
V. S. Urusov, Theoretical Crystallochemistry (Mosk. Gos. Univ., Moscow, 1987) [in Russian].
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.N. Eremin, R.A. Talis, V.S. Urusov, 2008, published in Kristallografiya, 2008, Vol. 53, No. 5, pp. 802–810.
Rights and permissions
About this article
Cite this article
Eremin, N.N., Talis, R.A. & Urusov, V.S. Computer modeling of the local structure, mixing properties, and stability of binary oxide solid solutions with corundum structure. Crystallogr. Rep. 53, 755–763 (2008). https://doi.org/10.1134/S1063774508050052
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1063774508050052