Abstract
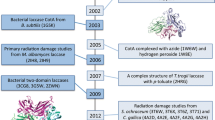
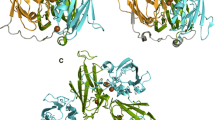
“Blue” copper-containing proteins are multidomain proteins that utilize a unique redox property of copper ions. Among other blue multicopper oxidases, three-domain oxidases belong to the group of proteins that exhibit a wide variety of compositions in amino acid sequences, functions, and occurrences in organisms. This paper presents a review of the data obtained from X-ray diffraction investigations of the three-dimensional structures of three-domain multicopper oxidases, such as the ascorbate oxidase catalyzing oxidation of ascorbate to dehydroascorbate and its three derivatives; the multicopper oxidase CueO (the laccase homologue); the laccases isolated from the basidiomycetes Coprinus cinereus, Trametes versicolor, Coriolus zonatus, Cerrena maxima, and Rigidoporus lignosus and the ascomycete Melanocarpus albomyces; and the bacterial laccases CotA from the endospore coats of Bacillus subtilis. A comparison of the molecular structures of the laccases of different origins demonstrates that, structurally, these objects are highly conservative. This obviously indicates that the catalytic activity of the enzymes under consideration is characterized by similar mechanisms.
Similar content being viewed by others
References
D. R. McMillin and M. K. Eggleston, in Multi-Copper Oxidases, Ed. by A. Messerschmidt (World Scientific, Singapore, 1997), Chapter 5, p. 129.
L. G. Ryden and L. T. Hunt, J. Mol. Evol. 36, 41 (1993).
K. Nakamura and N. Go, Cell. Mol. Life Sci. 62(18), 2050 (2005); (Birkhüser, Basel, 2005), p. 1.
E. T. Adman and M. E. P. Murphy, in Handbook of Metalloproteins, Ed. by A. Messerschmidt, R. Huber, T. Poulos, and K. Wieghardt (Wiley, New York, 2001), Vol. 2, p. 1381.
K. Nakamura, T. Kawabata, K. Yura, and N. Go, FEBS Lett. 553, 239 (2003).
R. Malkin and B. G. Malmström, Adv. Enzymol. Relat. Areas Mol. Biol. 33, 177 (1970).
B. Reinhammar, Copper Proteins Copper Enzymes 3, 1 (1984).
E. I. Solomon, M. J. Baldwin, and M. D. Lowery, Chem. Rev. 92, 521 (1992).
W. Kaim and J. Rall, Angew. Chem., Int. Ed. Engl. 35, 43 (1996).
M. D. Allendorf, D. J. Spira, and E. I. Solomon, Proc. Natl. Acad. Sci. USA 82, 3063 (1985).
E. Agostinelli, L. Cervoni, A. Giartosio, and L. Morpurgo, Biochem. J. 306, 697 (1995).
B. G. Malmström, Arch. Biochem. Biophys. 280, 233 (1990).
J. A. Farrar, A. J. Thomson, M. R. Cheesman, et al., FEBS Lett. 294, 11 (1991).
R. M. Wynn, H. K. Sarkar, R. A. Holwerda, and D. B. Knaff, FEBS Lett. 156, 23 (1983).
E. I. Solomon, U. M. Sundaram, and T. E. Machonkin, Chem. Rev. 96, 2563 (1996).
N. T. Dittmer, R. J. Suderman, H. Jiang, et al., Insect Biochem. Mol. Biol. 34, 29 (2004).
N. M. Parkinson, C. M. Conyers, J. N. Keen, et al., Comp. Biochem. Physiol., Part C: Toxicol. Pharmacol. 134, 513 (2003).
G. Alexandre and I. B. Zhulin, Trends Biotechnol. 18, 41 (2000).
H. Claus, Micron 35, 93 (2004).
H. Claus, Arch. Microbiol. 179, 145 (2003).
G. A. White and R. M. Krupka, Arch. Biochem. Biophys. 110, 448 (1965).
B. Mondovi, O. Befani, and S. Sabatini, Agents Actions 14, 356 (1984).
H. Itoh, A. Hirota, K. Hirayama, et al., Biosci., Biotechnol., Biochem. 59, 1052 (1995).
T. Nakamura, N. Makino, and Y. Ogura, J. Biochem. (Tokyo, Jpn.) 64, 189 (1968).
A. Marchesini and P. M. H. Kroneck, Eur. J. Biochem. 101, 65 (1979).
L. Avigliano, P. Vecchini, P. Sirianni, et al., Mol. Cell. Biochem. 56, 107 (1983).
M. H. Thuesen, O. Farver, B. Reinhammar, and J. Ulstrup, Acta Chem. Scand. 52, 555 (1998).
A. Messerschmidt, in Handbook of Metalloproteins, Ed. by A. Messerschmidt, R. Huber, T. Poulos, and K. Wieghardt (Wiley, New York, 2001), Vol. 2, p. 1345.
C. Pignocchi, J. M. Fletcher, J. E. Wilkinson, et al., Plant Physiol. 132, 1631 (2003).
H. Asao, K. Yoshida, Y. Nishi, and A. Shinmyo, Biosci., Biotechnol., Biochem. 67, 271 (2003).
M. A. Green and S. C. Fry, Nature (London) 433, 83 (2005).
A. Messerschmidt, R. Ladenstein, R. Huber, et al., J. Mol. Biol. 224, 179 (1992).
A. Messerschmidt, A. Rossi, R. Ladenstein, et al., J. Mol. Biol. 206, 513 (1989).
A. Messerschmidt, H. Luecke, and R. Huber, J. Mol. Biol. 230, 997 (1993).
A. Messerschmidt, R. Huber, and T. Poulos, in Handbook of Metalloproteins, Ed. by A. Messerschmidt, R. Huber, T. Poulos, and K. Wieghardt (Wiley, New York, 2001).
D. J. Winge, Adv. Protein Chem. 60, 51 (2002).
I. Voskoboinik, J. Camakaris, and J. F. B. Mercer, Adv. Protein Chem. 60, 123 (2002).
L. Banci, and A. Rosato, Acc. Chem. Res. 36, 215 (2003).
Z. H. Lu and M. Solioz, Adv. Protein Chem. 60, 93 (2002).
L. A. Finney and T. V. O’Halloran, Science (Washington) 300, 931 (2003).
C. Rensing and G. Grass, FEMS Microbiol. Rev. 27, 197 (2003).
M. Solioz and J. V. Stoyanov, FEMS Microbiol. Rev. 27, 183 (2003).
J. S. Cavet, G. P. Borrelly, and N. J. Robinson, FEMS Microbiol. Rev. 27, 165 (2003).
J. de Freitas, H. Wintz, J. H. Kim, et al., BioMetals 16, 185 (2003).
S. K. Singh, G. Grass, C. Rensing, and W. R. Montfort, J. Bacteriol. 186, 7815 (2004).
S. A. Roberts, A. Weichsel, G. Grass, et al., Proc. Natl. Acad. Sci. USA 99, 2766 (2002).
F. W. Outten, D. L. Huffman, J. A. Hale, and T. V. O’Halloran, J. Biol. Chem. 276, 30 670 (2001).
S. A. Roberts, G. F. Wildner, G. Grass, et al., J. Biol. Chem. 278(34), 31 958 (2003).
D. R. McMillin and M. K. Eggleston, in Multi-Copper Oxidases, Ed. by A. Messerschmidt (World Scientific, Singapore, 1997), Chapter 5, p. 129.
B. Reinhammar, in Copper Proteins and Copper Enzymes, Ed. by R. Lontie (CRC, Boca Raton, FL, United States, 1984), Vol. 3, p. 1.
A. I. Yaropolov, O. V. Skorobogat’ko, S. S. Vartanov, and S. D. Varfolomeev, Appl. Biochem. Biotechnol. 49(3), 257 (1994).
E. Torres and I. Bustos-Jaimes, Appl. Catal., B 46(1), 1 (2003).
H. Claus, G. Faber, and H. König, Appl. Microbiol. Biotechnol. 59(6), 672 (2002).
A. Leonowicz, Cho Nam-Seok, J. Luterek, J. Basic Microbiol. 41,(3–4), 185 (2001).
P. Bajpai, Biotechnol. Prog. 15, 147 (1999).
S. Shleev, J. Tkac, A. Chistenson, et al., Biosens. Bioelectron. 20, 2517 (2005).
A. Christenson, N. Dimcheva, E. E. Ferapontova, et al., Electroanalysis 16(13–14), 1074 (2004).
S. Shleev, J. Tkac, A. Christenson, et al., Biosens. Bioelectron. 20(12), 2517 (2005).
V. Ducros, A. M. Brzozowski, K. S. Wilson, et al., Nat. Struct. Biol. 5, 310 (1998).
V. Ducros, A. M. Brzozowski, K. S. Wilson, et al., Acta Crystallogr., Sect. D: Biol. Crystallogr. 57, 333 (2001).
K. Piontec, M. Antorini, and T. Choinowski, J. Biol. Chem. 277, 37663 (2002).
A. V. Lyashenko, N. E. Zhukhlistova, E. V. Stepanova, et al., Kristallografiya 51(2), 305 (2006) [Crystallogr. Rep. 51 (2), 278 (2006)].
A. V. Lyashenko, Yu. N. Zhukova, N. E. Zhukhlistova, et al., Kristallografiya 51(5), 870 (2006) [Crystallogr. Rep. 51 (5), 817 (2006)].
A. V. Lyashenko, I. Bento, V. N. Zaitsev, et al., JBIC, J. Biol. Inorg. Chem. 11, 963 (2006).
N. Hakulinen, L. L. Kiiskinen, K. Kruus, et al., Nat. Struct. Biol. 9, 601 (2002).
F. J. Enguita, L. O. Martins, A. O. Henriques, and M. A. Carrondo, J. Biol. Chem. 278, 19 416 (2003).
F. J. Enguita, D. Marcal, L. O. Martins, et al., J. Biol. Chem. 279, 23 472 (2004).
I. Bento, L. O. Martins, G. G. Lopes, et al., Dalton Trans. 21, 3507 (2005).
P. M. Coll, J. M. Fernandez-Abalos, J. R. Villanueva, et al., Appl. Environ. Microbiol. 59, 2607 (1993).
F. Xu, Biochemistry 35, 7608 (1996).
J. Sealey and A. J. Ragauskas, Enzyme Microb. Technol. 23, 422 (1998).
S. Garavaglia, M. T. Cambria, M. Miglio, et al., J. Mol. Biol. 342, 1519 (2004).
M. E. P. Murphy, P. F. Lindley, and E. T. Adman, Protein Sci. 6, 761 (1997).
T. E. Machonkin, L. Quintanar, A. E. Palmer, et al., J. Am. Chem. Soc. 123, 5507 (2001).
B. G. Malmström, B. Reinhammar, and T. Vanngard, Biochim. Biophys. Acta 205, 48 (1970).
F. Xu, R. M. Berka, J. A. Wahleithner, et al., Biochem. J. 334, 63 (1998).
R. Zilhao, M. Serrano, R. Isticato, et al., J. Bacteriol. 186, 1110 (2004).
M. Serrano, R. Zilhao, E. Ricca, et al., J. Bacteriol. 181, 3632 (1999).
L. O. Martins, C. M. Soares, M. M. Pereira, et al., J. Biol. Chem. 277, 18 849 (2002).
T. Bertrand, C. Jolivalt, P. Briozzo, et al., Biochemistry 41, 7325 (2002).
R. Bourbonnais, D. Leech, and M. G. Paice, Biochim. Biophys. Acta 1379, 381 (1998).
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Boris Konstatinovich Vaĭnshteĭn
Original Russian Text © N.E. Zhukhlistova, Yu.N. Zhukova, A.V. Lyashenko, V.N. Zaĭtsev, A. M. Mikhaĭlov, 2008, published in Kristallografiya, 2008, Vol. 53, No. 1, pp. 92–110.
Translated by O. Borovik-Romanova
Rights and permissions
About this article
Cite this article
Zhukhlistova, N.E., Zhukova, Y.N., Lyashenko, A.V. et al. Three-dimensional organization of three-domain copper oxidases: A review. Crystallogr. Rep. 53, 92–109 (2008). https://doi.org/10.1134/S1063774508010124
Received:
Issue Date:
DOI: https://doi.org/10.1134/S1063774508010124