Abstract
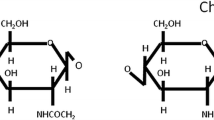
A series of four water-soluble chitosan derivatives differing in molecular mass, hydrophobicity, and charge was synthesized and tested for the intensity of their effects on Gram-negative and Gram-positive bacteria. It was shown that the tested compounds allowed the penetration of ethidium bromide into the bacteria, which showed increased permeability of their cell walls under the effect of chitosans. The tolerance to various chitosan derivatives differed in Gram-negative and Gram-positive bacteria. The Gram-negative bacteria were the most responsive to high-molecular chitosan and the Gram-positive ones, to N-,O-carboxypropylchitosan, whereas high-molecular chitosan had little effect. Research on the correlation between the structure and activity of the studied compounds revealed that depolymerization of chitosan reduced, and introduction of hydrophobic substantives in chitosan molecule significantly enhanced its permeability effect on bacterial cell walls. The obtained results provide a basis for the construction of new chitosan derivatives with antimicrobial activities.
Similar content being viewed by others
References
Bakholdina, S.I., Krasikova, I.N., Shubin F.N., et al., Effect of Culturing Technique and Growth Phase on Lipid Structure Yersinia pseudotuberculosis, Biokhimya, 2001, vol. 66, pp. 511–519.
Gerasimenko, D.V., Avdeenko, I.D., Bannikov, G.E., et al, Antibacterial Activity of Water-Soluble Low-Molecular Chitosans Concerning Various Microorganisms, Prikl. biokhimiya i mikrobiol., 2004, vol. 40, pp. 301–306.
Govorunov, I.G., Kosarev, N.V., Evtodienko, J.V., et al., Research of Permeability of Membranes of Cell Wall of Escherichia coli, Mikrobiologiya, 1982, vol. 51, no. 5, pp. 731–734.
Ivanushko, L.A., Soloveva, T.F., Zaporozhets, T.S., et al., Comparative Study of Immunomodulatory Properties of Chitosan and Its Derivatives, Med. immunol., 2007, vol. 9, no. 4–5, pp. 397–404.
Naberezhnykh, G.A., Gorbach, V.I., Likhatskaya, G.N., et al., Interaction of Chitosans and N-acylated Chitosan Derivatives with Lipopolysaccharides of Gram-Negative Bacteria, Biokhimiya, 2008, vol. 73, no. 4, pp. 530–541.
Nemtsev, S.V., A Complex Technology of Chitin and Chitosan Production from Crustaceans Carapaces, Moscow: VNIRO, 2006, pp. 7–24.
Nud’ga, L.M., Chitin and Chitosan Derivatives, Khitin i khitozan (Chitin and Chitosan), Moscow: Nauka, 2002, pp. 141–176.
Bengoechea, J.A., Diiaz, R., and Moriyon, I. Outer Membrane Differences between Pathogenic and Environmental Yersinia enterocolitica Biogroups Probed with Hydrophobic Permeants and Polycationic Peptides, Infec. Immun., 1996, vol. 64, no. 12, pp. 4891–4899.
Fischer, W., Lipoteichoic Acid and Lipids in the Membrane of Staphylococcus aureus, Med. Microbiol. Immunol., 1994, vol. 183, pp. 61–76.
Helander, I.M., Nurmiaho-Lassila, E.L., Ahvenainen, C., et al., Chitosan Disrupts the Barrier Properties of the Outer Membrane of Gram-Negative Bacteria, Int. J. Food Microbiol., 2001, vol. 71, pp. 235–244.
Je, J.Y., Kim, S.K., Chitosan Derivatives Killed Bacteria by Disrupting the Outer and Inner Membrane, J. Agric. Food Chem., 2006, vol. 54, pp. 6629–6633.
Jeon, Y.J. and Kim, S.K., Production of Chitooligosaccharides Using an Ultrafiltration Membrane Reactor and Their Antibacterial Activity, Carbohydr. Polym., 2000, vol. 41, pp. 133–144.
Kurita, K., Kojima, T., Nishiyama, Y., et al. Synthesis and Some Properties of Nonnatural Amino Polysaccharides: Branched Chitin and Chitosan, Macromolecule, 2000, vol. 79, pp. 4711–4716.
Liu, X.F., Guan, Y.L., Yang, D., et al., Antibacterial Action of Chitosan and Carboxymethylated Chitosan, J. Appl. Polym. Sci., 2001, vol. 79, pp. 1324–1335.
Lugtenberg, B. and Van Alphen, L., Molecular Architecture and Functioning of the Outer Membrane of Escherichia coli and other Gram-Negative Bacteria, Biochim. Biophys. Acta, 1983, vol. 737, no. 1, pp. 51–115.
Muzzarelli, R., Tarsi, R., Filippini, O., et al., Antimicrobial Properties of N-Carboxylbutyl Chitosan, Antimicrob. Agents Chemother., 1990, vol. 34, pp. 2019–2023.
Nikaido, H. and Vaara, M., Molecular Basis of Bacterial Outer Membrane Permeability, Microbiol. Rev., 1985, vol. 49, pp. 1–32.
No, K.H., Park, N.Y., Lee, S.H., et al., Antibacterial Activity of Chitosans and Chitosan Oligomers with Different Molecular Weights, Int. J. Food Microbiol., 2002, vol. 74, pp. 65–72.
Raafat, D., Bargen, K., Haas, A., et al., Insights into the Mode of Action of Chitosan as an Antibacterial Compound, Appl. Environ. Microbiol., 2008, vol. 74, pp. 3764–3773.
Rabea, E.I., Badawy, M.E.T., Stevens, C.V., et al., Chitosan as Antimicrobial Agent: Applications and Mode of Action, Biomacromolecules, 2003, vol. 4, pp. 1457–1465.
Singla, A.K. and Chawla, M., Chitosan: Some Pharmaceutical and Biological Aspects—An Update, J. Pharm. Pharmacol., 2001, vol. 53, pp. 1047–1067.
Tsai, C.-M. and Frasch, C.E., A Sensitive Silver Stain for Detecting Lipopolysaccharides in Polyacrylamide Gels, Anal. Biochem., 1982, vol. 119, no. 1, pp. 115–119.
Vaara, M. and Vaara, T., Polycations as Outer Membrane-Disorganizing Agents, Antimicrob. Agents Chemother., 1983, vol. 24, pp. 114–122.
Xie, W., Xu, P., Wang, W., et al., Preparation and Antibacterial Activity of a Water-Soluble Chitosan Derivative, Carbohydr. Polym., 2002, vol. 79, pp. 35–40.
Zorko, M., Majerle, A., Sarlah, D., et al., Combination of Antimicrobial and Endotoxin-Neutralizing Activities of Novel Oleoylamines, Antimicrob. Agents Chemother., 2005, vol. 49, pp. 2307–2313.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © G.A. Naberezhnykh, S.I. Bakholdina, V. I. Gorbach, T.F. Solov’eva, 2009, published in Biologiya Morya.
Rights and permissions
About this article
Cite this article
Naberezhnykh, G.A., Bakholdina, S.I., Gorbach, V.I. et al. New chitosan derivatives with potential antimicrobial activity. Russ J Mar Biol 35, 498–503 (2009). https://doi.org/10.1134/S106307400906008X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S106307400906008X