Abstract
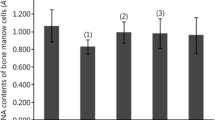
In experiments in mice, the radioprotective efficacy of sugar-containing compounds was studied based on the condensation products of thioglycolic (TGH) and thiosalicylic (TSH) hydrazides with a series of natural aldoses: the monosaccharides D-glucose (Glс) and D-galactose (Gal) and the disaccharides D‑lactose (Lac) and D-maltose (Mal). It was shown that the use of Glc-TGH and Mal-TSH preparations increases the survival rate of deadly irradiated animals by 25% when administered 1 h before irradiation and by 40% when administered 24 h before irradiation. It is assumed that the leading role in the radioprotective activity of the drugs is played by the protection of the hematopoietic stem cells of the test animals.


Similar content being viewed by others
REFERENCES
García, I., Gallo, J., Marradi, M., et al., Glyconanoparticles: new nanomaterials for biological application, in Engineered Carbohydrate-Based Materials for Biomedical Applications: Polymers, Surfaces, Dendrimers, Nanoparticles, and Hydrogels, Narain, R., Ed., New Jersey: Wiley, 2011, ch. 2, pp. 213–250.
Fedotcheva, T.A., Starostin, K.M., and Shimanovskii, N.L., Prospects for using gold, silver, and iron oxide nanoparticles for increasing the efficacy of chemotherapy, Pharm. Chem. J., 2015, vol. 49, no. 4, pp. 220–230. https://doi.org/10.1007/s11094-015-1260-6
Bhattarai, J.K., Neupane, D., Mikhaylov, V., et al., Self-assembled monolayers of carbohydrate derivatives on gold surfaces, in Carbohydrate, Caliskan, M., Kavakli, I.H., and Oz, G.C., Eds., InTech, 2017, ch. 4, pp. 63–97. https://doi.org/10.5772/66194
Haume, K., Rosa, S., Grellet, S., et al., Gold nanoparticles for cancer radiotherapy: a review, Cancer Nano, 2016, vol. 7/8, pp. e1–20. https://doi.org/10.1186/s12645-016-0021-x
Yang, C., Bromma, K., Di Ciano-Oliveira, C., et al., Gold nanoparticle mediated combined cancer therapy, Cancer Nano, 2018, vol. 9/4, pp. e1–14. https://doi.org/10.1186/s12645-018-0039-3
Johnke, R.M., Sattler, J.A., and Allison, R.R., Radioprotective agents for radiation therapy: future trends, Future Oncol., 2014, vol. 10, no. 15, pp. 2345–2357. https://doi.org/10.2217/fon.14.175
Ershov, A.Y., Vasil’eva, M.Y., Lagoda, I.V., et al., Condensation products of D-ribose with thiol-containing hydrazides and gold glyconanoparticles thereof, Russ. J. Gen. Chem., 2018, vol. 88, no. 1, pp. 103–108. https://doi.org/10.1134/S1070363218010164
Vasil’eva, M.Y., Ershov, A.Y., Baigil’din, V.A., et al., Synthesis of silver glyconanoparticles based on 3-thiopropionylhydrazones of mono- and disaccharides, Russ. J. Gen. Chem., 2018, vol. 88, no. 1, pp. 109–113. https://doi.org/10.1134/S1070363218010176
Ershov, A.Y., Vasil’eva, M.Y., Lagoda, I.V., et al., Structure of the condensation products of D-lactose and d-maltose with SH-containing carboxylic acid hydrazides, Russ. J. Gen. Chem., 2018, vol. 88, no. 6, pp. 1199–1204. https://doi.org/10.1134/S1070363218060245
Vasil’eva, M.Y., Ershov, A.Y., Baigil’din, V.A., et al., Synthesis of gold glyconanoparticles based on the condensation products of D-lactose and D-maltose with SH-containing hydrazides, Russ. J. Gen. Chem., 2018, vol. 88, no. 6, pp. 1205–1209. https://doi.org/10.1134/S1070363218060257
Ershov, A.Y., Martynenkov, A.A., Lagoda, I.V., et al., Synthesis of 4-[ω-(mercaptoalkyl)oxy]benzoylhydrazones of mono- and disaccharides as potential glycoligands of noble metal nanoparticles, Russ. J. Gen. Chem., 2019, vol. 89, no. 2, pp. 292–299. https://doi.org/10.1134/S1070363219020208
Ershov, A.Y., Vasil’eva, M.Y., Levit, M.L., et al., Synthesis of gold glyconanoparticles based on thiol-containing D-hexose acylhydrazones and their modification by thiolated poly(2-deoxy-2-methacryloylamino-D-glucose), Russ. J. Gen. Chem., 2019, vol. 89, no. 2, pp. 300–308. https://doi.org/10.1134/S107036321902021X
Mikhailov, P.P. and Korovkina, E.P., Methodological guidelines for the preclinical study of the radioprotective properties of pharmacological compounds, in Rukovodstvo po eksperimental’nomu (doklinicheskomu) izucheniyu novykh farmakologicheskikh veshchestv (Guidelines for Experimental (Preclinical) Study of New Pharmacological Compounds), Khabriev, R.U., Ed., 2nd ed., Moscow: Meditsina, 2005, pp. 724–429.
Metodicheskie rekomendatsii po voprosam opredeleniya chislennosti krovetvornykh kolonieobrazuyushchikh edinits (KOE) s pomoshch’yu testov ekzogennykh i endogennykh selezenochnykh kolonii (Methodological Guidelines on the Determination of the Number of Hematopoietic Colony-Forming Units (CFU) using Tests of Exogenous and Endogenous Spleen Colonies), Obninsk: MRNTs RAMN, 1975.
Berezovskaya, I.V., Classification of chemicals by parameters of acute toxicity with parenteral routes of administration, Khim.-Farm. Zh., 2003, vol. T. 37, no. 3, pp. 32–34.
Filimonova, M.V. and Filimonov, A.S., Modern problems and prospects for the development and implementation of domestic antiradiation drugs, Radiats. Biol. Radioekol., 2019, vol. 59, no. 2, pp. 127–131.
Singh, V., Newman, V.L., Romaine, P.L., et al., Radiation countermeasure agents: an update (2011–2014), Expert. Opin. Ther., vol. 24, no. 11, pp. 1229–1255.
Potučková, E., Hrušková, K., Bureš, J., et al., Structure-activity relationships of novel salicylaldehyde isonicotinoyl hydrazone (SIH) analogs: iron chelation, anti-oxidant and cytotoxic properties, PLoS One, 2014, vol. 9, no. 11. e112059. https://doi.org/10.1371/journal.pone.0112059
Ponka, P., Borova, J., Neuwirt, J., et al., Mobilization of iron from reticulocytes. identification of pyridoxal isonicotinoyl hydrazone as a new iron chelating agent, FEBS Lett., 1997, vol. 97, no. 2, pp. 317–321. https://doi.org/10.1016/0014-5793(79)80111-8
Caro, A.A., Commissariat, A., Dunn, C., et al., Prooxidant and antioxidant properties of salicylaldehyde isonicotinoyl hydrazone iron chelators in HepG2 cells, Biochim. Biophys. Acta, 2015, vol. 1850, no. 11, pp. 2256–2264. https://doi.org/10.1016/j.bbagen.2015.08.005
Charkoudian, L.K., Pham, D.M., and Franz, K.J., A pro-chelator triggered by hydrogen peroxide inhibits iron-promoted hydroxyl radical formation, J. Am. Chem. Soc., 2006, vol. 128, no. 38, pp. 12424–12425. https://doi.org/10.1021/ja064806w
Vladimirov, V.G., Krasilnikov, I.I., and Arapov, O.V., Radioprotektory: struktura i funktsiya (Radioprotectors: Structure and Function), Kiev: Naukova dumka, 1989.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest. The authors declare that they have no conflict of interest.
Statement on the welfare of animals. During the study, the requirements of regulatory legal acts on the procedure for experimental work using animals, including a humane attitude to them, were met: “Code of Ethics” (1985), including the section “International Recommendations for Biomedical Research Using Animals,” the Helsinki Declaration of the World Medical Association (2000), etc.
Rights and permissions
About this article
Cite this article
Lagoda, I.V., Yakunchikova, E.A., Drachyov, I.S. et al. Investigation of the Radioprotective Efficiency of Condensation Products of Thiol-Containing Hydrazides with Mono- and Disaccharides. Biol Bull Russ Acad Sci 47, 1680–1685 (2020). https://doi.org/10.1134/S1062359020120043
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1062359020120043