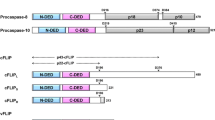
Abstract
The data of recent years on apoptosis were revisited to demonstrate that the functioning of signaling proteins during apoptosis depends on their localization on mitochondria or in the cytosol. The major effect of signaling proteins depends on the number of pro-and antiapoptotic domains in their structure, which is observed after cleavage, oligomerization, and complexing with other proteins. The structure of known signaling proteins was analyzed. The effect of complexing with phosphatases and 14-3-3 proteins was demonstrated by the example of Bad protein. Detailed data on the proapoptotic factors and their inhibitors affecting caspase activation and released from mitochondria with cytochrome c are given.
Similar content being viewed by others
References
Ayllon, V., Cayla X., Garsia A. et al. Bcl-2 Targets Protein Phosphatase 1 alpha to Bad, J. Immunol., 2001, vol. 166, no. 12, pp. 7345–7352.
Blink, E., Maianski, N.A., Alnemri, E.S., et al., Intramitochondrial Serine Protease Activity of Omi/HtrA2 Is Required for Caspase-Independent Cell Death of Human Neutrophils, Cell Death. Differ., 2004, vol. 11, pp. 937–939.
Bossy-Wetzel, E., Newmeyer, D.D., and Green, D.R., Mitochondrial Cytochrome C Release in Apoptosis Occurs upstream of DEVD-Specific Caspase Activation and Independently of Mitochondrial Transmembrane Depolarization, EMBO J., 1998, vol. 17, no. 1, pp. 37–49.
Bratton, S.B., Walker, G., Roberts, D.L., et al., Caspase-3 Cleaves Apaf-1 into an Appropriately Oligomerized and Biologically Inactive Approximately 1.5 MDa Apoptosome Complex, Cell Death Differ., 2001, vol. 8, no. 4, pp. 425–433.
Burgering, B.M. and Kops, G.J., Cell Cycle and Death Control: Long Life Forkheads, Trends Biochem. Sci., 2002, vol. 27, no. 5, pp. 352–360.
Cario, G., Skokowa, J., Wang, Z., et al., Heterogenous Expression Pattern of Pro-and Anti-Apoptotic Factors in Myeloid Progenitor Cells of Patients with Severe Congenital Neutropenia Treated with Granulocyte Stimulating Factor, Br. J. Haematol., 2005, vol. 129, no. 2, pp. 275–258.
Chai, J., Du, C., Wu, J.-W., et al., Structural and Biochemical Basis of Apoptotic Activation by SMAC/Diablo, Nature, 2000, vol. 406, no. 6798, pp. 855–862.
Chang, D.W., Zheng, X., Pan, Y., et al., C-FlipL Is a Dual Function Regulator for Caspase-8 Activation and CD95-Mediated Apoptosis, EMBO J., 2000, vol. 21, no. 14, pp. 3704–3714.
Chen, Q., Chai, Y., Mazumder, S., et al., Double Steps of Cytochrome C Appearance under ROS Increase, Sci. World J., 2001, vol. 1, no. 1, pp. 142–144.
Chiang, C.W., Harris, G., Ellig, C., et al., Protein Phosphatase 2A Activates the Proapoptotic Function of BAD in Interleukin-3-Dependent Lymphoid Cells by a Mechanism Requiring 14-3-3 Dissociation, Blood, 2001, vol. 97, no. 5, pp. 1289–1297.
Chiang, C.W., Kanies C., Kim K.W., et al., Protein Phosphatase 2A Dephosphorylation of Phosphoserine 112 Plays the Gatekeeper Role for BAD-Mediated Apoptosis, Mol. Cell. Biol., 2003, vol. 23, no. 18, pp. 6350–6362.
Clem, R.J., Cheng, E.H., Karp, C.L., et al., Modulation of Cell Death by Bcl-XL through Caspase Interaction, Proc. Natl. Acad. Sci. USA, 1998, vol. 95, no. 2, pp. 554–559.
Clem, R.J., Sheu, T.T., and Richter, B.W., C-IAPs Is Cleavled to Produce a Pro-Apoptitic C-Terminal Fragment, J. Biol. Chem., 2001, vol. 276, no. 10, pp. 7601–7608.
Cook, S.A., Novikov, M.S., Ahn, Y., et al., A20 Is Dynamically Regulated in the Heart and Inhibits the Hypertrophic Response, Circulation, 2003, vol. 108, no. 6, pp. 664–667.
Cowburn, A.S., Cadwallader, K.A., Reed, B.J., et al., Role of PI3-Kinase Bad Phosphorilation and Altered Transcription in Cytokine-Mediated Neutrophil Survival, Blood, 2002, vol. 100, no. 7, pp. 2607–2616.
Datta, S.R., Katsov, A., Hu, L., et al., 14-3-3 Proteins and Survival Kinases Cooperate to Inactivate BAD by BH3 Domain Phosphorylation, Mol. Cell, 2000, vol. 6, no. 1, pp. 41–51.
Datta, S.R., Ranger, A.M., Lin, M.Z., et al., Survival Factor-Mediated BAD Phosphorylation Raises the Mitochondrial Threshold for Apoptosis, Dev. Cell, 2002, vol. 3, no. 5, pp. 631–643.
Davies, S.P., Reddy, H., Caivano, M., and Cohen, P., Specificity and Mechanism of Action of Some Commonly Used Protein Kinase Inhibitors, Biochem. J., 2000, vol. 351, pp. 95–106.
Desagher, S., Osen-Sand, A., Montessuit, S., et al., Phosphorylation of Bid by Casein Kinases and Regulation Its Cleavage by Caspase 8, Mol. Cell, 2001, vol. 8, no. 3, pp. 601–611.
Deveraux, Q.L., Leo, E., and Stennicke, H.R., Cleavage of Human Inhibitor of Apoptosis Protein XIAP Results in Fragments with Distinct Specifities for Caspases, Blood, 1999, vol. 94, no. 10, pp. 3864–3870.
Domina, A.M., Smith, J.H., and Craig, R.W., Myeloid Cell Leukemia 1 Is Phosphorylated through Two Distinct Pathways, One Associated with Extracellular Signal-Regulated Kinase Activation and the Other with G2/M Accumulation or Protein Phosphatase 1/2A Inhibition, J. Biol. Chem., 2000, vol. 275, no. 28, pp. 21688–21694.
Dramsi, S., Scheid, M.P., Maiti, A., et al., Identification of a Novel Phosphorylation Site, Ser-170, as a Regulator of Bad Proapoptotic Activity, J. Biol. Chem., 2002, vol. 277, no. 8, pp. 6399–6405.
Du, C., Fang, M., Li, Y., and Wang, X., Smac, A Mitochondrial Protein That Promotes Cytochrome C-Dependent Caspase Activation by Eliminating IAP Inhibition, Cell, 2000, vol. 102, no. 1, pp. 33–42.
Edwards, S.W., Derouet, M., Howse, M., and Moots, R.J., Regulation of Neutrophil Apoptosis by Mcl-1, Biochem. Soc. Trans., 2004, vol. 32, no. 3, pp. 489–492.
Esposti, M.D., Erler, J.T., Hickman, J.A., and Dive, C., Bid, A Widely Expressed Proapoptotic Protein of the Bcl-2 Family, Displays Lipid Transfer Activity, Mol. Cell Biol., 2001, vol. 21, no. 21, pp. 268–276.
Esposti, M.D., Mitochondria in Apoptosis: Past, Present and Future, Biochem. Soc. Trans., 2004, vol. 32, pp. 493–495.
Fang, L.W., Tai, T.S., Yu, W.N., et al., Phosphatidylinositide 3-Kinase Priming Couples c-FLIP to T Cell Activation, J. Biol. Chem., 2004, vol. 279, no. 1, pp. 13–18.
Gajkowska, B. and Wojewodska, U., A Novel Embedment-Free Immunoelectron Microscopy Technique Reveals Association of Apoptosis-Regulating Proteins with Subcellular Structures, Histochem. J., 2002, vol. 34, nos. 8–9, pp. 441–446.
Hasegawa, T., Suzuki, K., Sakamoto, Ch., et al., Expression of the Inhibitor of Apoptosis (IAP) Family Members in Human Neutrophils: Up-Regulation of CIAP2 by Granulocyte Colony-Stimulating Factor and Overexpression of CIAP2 in Chronic Neutrophilic Leukemia, Blood, 2003, vol. 101, no. 3, pp. 1164–1171.
Haupt, S., Berger, M., Goldberg, Z., and Haupt, Y., Apoptosis—The p53 Network, J. Cell Sci., 2003, vol. 116, no. 20, pp. 4077–4085.
He, K.L. and Ting, A.T., A20 Inhibits Tumor Necrosis Factor (TNF) Alpha-Induced Apoptosis by Disrupting Recruitment of TRADD and RIP to the TNF Receptor 1 Complex in Jurkat T Cells, Mol. Cell Biol., 2002, vol. 22, no. 17, pp. 6034–6040.
Hirai, I. and Wang, H.G., Survival-Factor-Induced Phosphorylation of Bad Results in Its Dissociation from Bcl-XL but Not Bcl-2, Biochem. J., 2001, vol. 359, no. 2, pp. 345–352.
Hirotani, M., Zhang, Y., Fujita, N., et al., NH2-Terminal BH4 Domain of Bcl-2 Is Functional for Heterodimerization with Bax and Inhibition of Apoptosis, J. Biol. Chem., 1999, vol. 274, no. 29, pp. 20415–20420.
Huang, D.C. and Strasser, A., BH3-Only Proteins-Essential Initiators of Apoptotic Cell Death, Cell, 2000, vol. 103, no. 6, pp. 839–842.
Huang, Y., Rich, R.L., Myszka, D.G., and Wu, H., Requirement of Both the BIR2 and BIR3 Domains for the Relief of XIAP-Mediated Caspase Inhibition by Smac, J. Biol. Chem., 2003, vol. 278, no. 49, pp. 49517–49522.
Inoshita, S., Takeda, K., Hatai, T., et al., Phosphorilation and Inactivation of Myeloid Cell Leukemia 1 by JNK in Response to Oxidative Stress, J. Biol. Chem., 2002, vol. 277, no. 46, pp. 43730–43734.
Jonsson, G., Paulie, S., and Grandien, A., High Level of CFLIP Correlates with Resistance to Death Receptor-Induced Apoptosis in Bladder Carcinoma Cells, Anticancer Res., 2003, vol. 23, no. 2, pp. 1213–1218.
Kang, C.M., Suh, Y., Jang, I.S., and Park, S.C., Thymidine-Dependent Attenuation of the Mitochondrial Apoptotic Pathway in Adenosine-Induced Apoptosis of HL-60 Cells, J. Cancer Res. Clin. Oncol., 2001, vol. 127, no. 9, pp. 570–576.
Kerr, J.F.R., Wyllie, A.H., and Currie, A.R., Apoptosis: A Basic Biological Phenomenon with Wide-Ranging Implications in Tissue Kinetics, Br. J. Cancer, 1972, vol. 26, no. 4, pp. 239–257.
Ko, J.K., Choi, K.H., Kim, H.J., et al., Conversion of Bfl-1, An Anti-Apoptotic Bcl-2 Family Protein, to a Potent Pro-Apoptotic Protein by Fusion with Green Fluorescent Protein (GFP), FEBS Lett., 2003, vol. 551, nos. 1–3, pp. 29–36.
Konishi, Y., Lehtinen, M., Donovan, N., and Bonni, A., Cdc2 Phosphorylation of BAD Links the Cell Cycle to the Cell Death Machinery, Mol. Cell, 2002, vol. 9, no. 5, pp. 1005–1016.
Leblanc, V., Dery, M.C., Shooner, C., and Asselin, E., Opposite Regulation of XIAP and Smac/DIABLO in the Rat Endometrium in Response to 17beta-Estradiol at Estrus, Reprod. Biol. Endocrinol., 2003, vol. 1, no. 1, pp. 59–69.
Lorick, K.L., Jensen, J.P., Fang, S., et al., RING Fingers Mediate Ubiquitin-Conjugating Enzime E2-Dependent Ubiquitination, Proc. Natl. Acad. Sci. USA, 1999, vol. 96, no. 20, pp. 11364–11369.
Luetjens, C.M., Kogel, D., Reimertz, C., et al., Multiple Kinetics of Mitochondrial Cytochrome c Release in Drug-Induced Apoptosis, Mol. Pharmacol., 2001, vol. 60, no. 5, pp. 1008–1019.
Maianski, N.A., Roos, D., and Kuijpers, T.W., Tumor Necrosis Factor Alpha Induces a Caspase-Independent Death Pathway in Human Neutrophils, Blood, 2003, vol. 101, no. 5, pp. 1987–1995.
Malewicz, M., Zeller, N., Yilmaz, Z.B., and Weih, F., NF-Kappa B Controls the Balance Between Fas and Tumor Necrosis Factor Cell Death Pathways during T Cell Receptor-Induced Apoptosis via the Expression of Its Target Gene A20, J. Biol. Chem., 2003, vol. 278, no. 35, pp. 32825–32833.
Martins, L.M., The Serine Protease Omi/HtrA2: A Second Mammalian Protein with a Reaper-like Function, Cell Death Differ., 2002, vol. 9, no. 7, pp. 699–701.
Masters, S.C., Yang, H., Datta, S.R., et al., 14-3-3 Inhibits Bad-Induced Cell Death through Interaction with Serine-136, Mol. Pharmacol., 2001, vol. 60, no. 6, pp. 1325–1331.
Masters, S.C., Subramanian, R.R., Truong, A., et al., Survival-Promoting Functions of 14-3-3 Proteins, Biochem. Soc. Trans., 2002, vol. 30, no. 4, pp. 360–365.
Mayanskii, A.N., Mayanskii, N.A., Zaslavskaya, N.I., et al., Apoptosis of Neutrophils, Immunologiya, 1999, vol. 20, no. 6, pp. 11–20.
Michels, J., Johson, P.W., and Packman, G., Mcl-1, Int. J. Biochem. Cell Biol., 2005, vol. 37, no. 2, pp. 267–271.
Mortenson, M.M., Schlieman, M.G., Virudalchalam, S., and Bold, R.J., Overexpression of Bcl-2 Results in Activation of the Akt/NF-KB Cell Survival Pathway, J. Surg. Res., 2003, vol. 114, no. 2, pp. 302–310.
Nicholson, D.W., Baiting Death Inhibitor, Nature, 2001, vol. 410, no. 6824, pp. 33–34.
Opferman, J.T., Letai, A., Beard, C., et al., Development and Maintenance of T and B Lymphocytes Requires Antiapoptotic Mcl-1, Nature, 2003, vol. 426, no. 6967, pp. 671–674.
Pratt, M.A.C., Niu, M.-Y., Bcl-2 Controls Caspase Activation Following a p53-Dependent Cyclin D1-Induced Death Signal, J. Biol. Chem., 2003, vol. 278, no. 18, pp. 14211–14229.
Rehm, M., Dussmann, H., and Prehn, J.H., Real-Time Single Cell Analysis of Smac/DIABLO Release during Apoptosis, J. Cell Biol., 2003, vol. 162, no. 6, pp. 1031–1043.
Rodriguez, J. and Lazebnik, Y., Caspase-9 and Apaf-1 Form an Active Holoenzyme, Genes Dev., 1999, vol. 13, no. 24, pp. 3179–3184.
Rosa, J., Canovas, P., and Islam, A., Survivin Modulates Microtubule Dynamics and Nucleation throughout the Cell Cycle, Mol. Biol. Cell, 2006, vol. 17, no. 3, pp. 1483–1493.
Ruvolo, P.P., Deng, X., Ito, T., et al., Ceramide Induces Bcl-2 Dephosphorilation via a Mechanism Involving Mitochondrial PP2A, J. Biol. Chem., 1999, vol. 274, no. 29, pp. 20296–20300.
Sabbatini, P. and McCormik, F., PI3k and PKB/Akt Delay Onset of p53-Mediated Trascriptionally Dependent Apoptosis, J. Biol. Chem., 1999, vol. 274, no. 34, pp. 24263–24269.
Saito, A., Hayashi, T., Okuno, S., et al., Interaction between XIAP and Smac/DIABLO in the Mouse Brain after Transient Focal Cerebral Ischemia, J. Cereb. Blood Flow Metab., 2003, vol. 23, no. 9, pp. 1010–1019.
Samuilov, A.D., Oleskin, O.V., and Lagunova, E.M., Programmed Cell Death, Biokhimiya, 2000, vol. 65, no. 8, pp. 1029–1045.
Sano, J., Oguma, K., Kano, R., et al., High Expression of Bcl-xL in Delayed Apoptosis of Canine Neutrophils Induced by Lipopolysaccharide, Res. Vet. Sci., 2005, vol. 78, no. 2, pp. 183–187.
Sanz, C., Benito, A., Silva, M., et al., The Expression of Bcl-XL Is Downregulated during Differentiation of Human Hematopoietic Progenitor Cells along the Granulocyte but Not the Monocyte/Macrophage Lineage, Blood, 1997, vol. 89, no. 9, pp. 2199–2204.
Schimmer, A.D., Hedley, D.W., Pham, N.A., et al., BAD Induces Apoptosis in Cells Over-Expressing Bcl-2 or Bcl-XL without Loss of Mitochondrial Membrane Potential, Leuk. Lymphoma, 2001, vol. 42, no. 3, pp. 429–443.
Schurmann, A., Mooney, A.F., Sanders, L.C., et al., p21-Activated Kinase 1 Phosphorylates the Death Agonist Bad and Protects Cells from Apoptosis, Mol. Cell. Biol., 2000, vol. 20, no. 2, pp. 453–461.
Simmons, T.D., Slater, K.J., and Crouch, S.P., Changes in Intracellular ADP: ATP Ratios as a Marker of Apoptosis, Sci. World J., 2001, vol. 1, no. Suppl. 3, p. 58.
Slee, E.A., Hartle, M.T., Kluck, R.M., et al., Ordering the Cytochrome C-Initiated Caspase Cascade: Hierarchical Activation of Caspase-2,-3,-6,-7,-8 and-10 in a Caspase-9-Dependent Manner, J. Cell Biol., 1999, vol. 144, no. 2, pp. 281–292.
Srinivasula, R.K., Mi, Q.S., Hardwick, J.M., et al., Deletion of the Loop Region of Bcl-2 Completely Blocks Paclitaxel-Induced Apoptosis, Proc. Natl. Acad. Sci. USA, 1999, vol. 96, pp. 3775–3780.
Srinivasula, S.M., Hedge, R., and Saleh, A., A Conserved XIAP-Interaction Motif in Caspase 9 and Smac / Diablo Regulates Caspase Activity and Apoptosis, Nature, 2001, vol. 410, no. 6824, pp. 112–116.
Srinivasula, S.M., Gupta, S., Datta, P., et al., Inhibitors of Apoptosis Proteins Are Substrates for the Mitochondrial Serine Protease Omi/HtrA2, J. Biol. Chem., 2003, vol. 278, no. 34, pp. 31469–31472.
Sundararajan, R., Cuconati, A., Nelson, D., and White, E., TNF-Alpha Induces Bax-Bak Interaction and Apoptosis Which Is Inhibited by Adenovirus E1B 19K, J. Biol. Chem., 2001, vol. 276, no. 48, pp. 45120–45127.
Suzuki, Y., Imai, Y., Nakayama, H., et al., A Serine Protease, HtrA2, Is Released from the Mitochondria and Interacts with XIAP, Inducing Cell Death, Mol. Cell, 2001, vol. 8, no. 3, pp. 613–621.
Tadros, A., Hughes, D.P., Dunmore, B.J., and Brindle, N.P., ABIN-2 Protects Endothelial Cells from Death and Has a Role in the Antiapoptotic Effect of Angiopoietin-1, Blood, 2003, vol. 102, no. 13, pp. 4407–4409.
Tan, K., Tan, K.M., and Yu, V., A Novel BH3-Like Domain in BID Is Required for Intramolecular Interaction and Auto-inhibition of Pro-Apoptotic Activity, J. Biol. Chem., 1999, vol. 274, no. 34, pp. 23687–23690.
Tan, Y., Demeter, M.R., Ruan, H., and Comb, M.J., BAD Ser-155 Phosphorylation Regulates BAD/Bcl-XL Interaction and Cell Survival, J. Biol. Chem., 2000, vol. 275, no. 33, pp. 25865–25869.
Terradillos, O., Montessuit, S., Huang, D.C., and Martinou, J.C., Direct Addition of BimL to Mitochondria Does Not Lead to Cytochrome C Release, FEBS Lett., 2002, vol. 522, nos. 1–3, pp. 29–34.
Townsend, P.A., Stephanou, A., Packham, G., and Latchman, D.S., Bag-1: A Multifunctional Prosurvival Molecule, Int. J. Biochem. Cell Biol., 2005, vol. 37, no. 2, pp. 251–259.
Tyazhelova, V.G., Mekhanizmy aktivatsii limfotsitov perifericheskoi krovi (The Mechanisms of Peripheral Lymphocyte Activation), Moscow: Nauka, 2003a.
Tyazhelova, V.G., Pro-and Antiapoptotic Factors during the Activation of Peripheral Lymphocytes, Uspekhi Biol. Nauk, 2003b, no. 5, pp. 495–505.
Tyazhelova, V.G., TRAIL-and WNT-Signaling in Apoptosis, Immunologiya, 2005, vol. 26, no. 6, pp. 377–384.
Valentijn, A.J., Zouq, N., and Glimore, A.P., Anoikis, Biochem. Soc. Trans., 2004, vol. 32, no. 3, pp. 421–425.
Vaux, D.L. and Krosmeyer, S.J., Cell Death in Development, Cell, 1999, vol. 96, no. 2, pp. 243–254.
Verhagen, A.M. and Vaux, D.L., Cell Death Regulation by the IAP Antagonist Diablo/Smac, Apoptosis, 2000, vol. 7, no. 2, pp. 163–166.
Verhagen, A.M., Ekert, P.G., Pakusch, M., et al., Identification of Diablo, A Mammalian Protein That Promotes Apoptosis by Binding to and Antagonizing IAP Proteins, Cell, 2000, vol. 102, no. 1, pp. 43–53.
Wang, H.G. and Reed, J.S., Bcl-2, Raf-1 and Mitochondrial Apoptosis, Histol. Histopathotol., 1998, vol. 13, no. 2, pp. 521–530.
Wang, J.-M., Ming-Zong, L., and Hsin-Fang, Y.-Y., Interleukin-3 Stimulation of mcl-1 Gene Transcription Involves Activation of the PU.1 Transcription Factor through a p38 Mitogen-Activated Protein Kinase-Dependent Pathway, Mol. Cell Biol., 2003, vol. 23, no. 6, pp. 1896–1909.
Wang, N.S., Unkila, M.T., Reineks, E.Z., and Distelhorst, C.W., Transient Expression of Wild-Type or Mitochondrially Targeted Bcl-2 Induces Apoptosis while Transient Expression of ER-Targeted Bcl-2 Is Protective against Bax Induced Cell Death, J. Biol. Chem., 2001, vol. 276, no. 47, pp. 44117–44148.
Wilker, E. and Yaffe, M.B., 14-3-3 Proteins—A Focus on Cancer and Human Disease, J. Mol. Cell, 2004, vol. 37, no. 3, pp. 633–642.
Willie, A.H., Kerr, J.F., and Currie, A.R., Cell Death: The Significance of Apoptosis, Int. Rev. Cytol., 1980, vol. 68, pp. 251–306.
Wills, S.N. and Adams, J.M., Life in the Balance: How BH3 Only Proteins Induce Apoptosis, Curr. Opin. Cell Biol., 2005, vol. 17, no. 6, pp. 617–625.
Wu, Ch., Chai, J., Suber, T., et al., Structural Basis of IAP Recognition by Smac/Diablo, Nature, 2000, vol. 408, no. 6815, pp. 1008–1012.
Yang, Y., Fang, S., Jensen, J.P., et al., Ubiquitin Protein Ligase Activity of IAPs and Their Degradation in Proteasomes in Response to Apoptotic Stimuli, Science, 2000, vol. 288, no. 5467, pp. 874–877.
Zhou, X.M., Liu, Y., Payne, G., et al., Growth Factors Inactivate the Cell Death Promoter BAD by Phosphorylation of Its BH3 Domain on Ser155, J. Biol. Chem., 2000, vol. 275, no. 32, pp. 25046–25051.
Author information
Authors and Affiliations
Additional information
Original Russian Text © V.G. Tyazhelova, 2007, published in Izvestiya Akademii Nauk, Seriya Biologicheskaya, 2007, No. 2, pp. 133–144.
Rights and permissions
About this article
Cite this article
Tyazhelova, V.G. The role of the interaction between signaling protein domains and of the complexes of signaling proteins in apoptosis initiation. Biol Bull Russ Acad Sci 34, 99–109 (2007). https://doi.org/10.1134/S106235900702001X
Received:
Issue Date:
DOI: https://doi.org/10.1134/S106235900702001X