Abstract
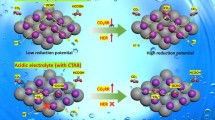
The composite materials involving platinum nanoparticles on the surface of carbides of Mo, Zr, and Nb are synthesized by the currentless redox reaction between potassium tetrachloroplatinate(II) and the corresponding carbide (supporting electrolyte—0.5 М H2SO4). The greatest amount of platinum is deposited in the reduction of tetrachloroplatinate(II) by molybdenum carbide Mo2C, and the smallest amount is deposited in the reduction by ZrC. The synthesized materials are characterized by the methods of scanning electron microscopy (SEM), X-ray photoelectron spectroscopy (XPS), X-ray diffraction (XRD), and cyclic voltammetry. The specific surface area of platinum deposited from solutions containing 0.05 M platinum salt during the deposition time of 20 min is found to be 28 m2/g for Pt/Mo2C, 26 m2/g for Pt/ZrC, and 10 m2/g for Pt/NbC. These materials exhibit the high catalytic activity in the reaction of electrochemical hydrogen evolution in acidic solutions.










Similar content being viewed by others
Notes
The electrode potentials of half-reactions (4) and (5) were calculated based on the standard Gibbs energies of formation of substances shown in [39].
REFERENCES
Naimi, Y. and Antar, A., Hydrogen generation by water electrolysis, in Advances in Hydrogen Generation Technologies, Eyvaz, M., Ed., IntechOpen, 2018.https://doi.org/10.5772/intechopen.76814
Bagotzky, V.S., Osetrova, N.V., and Skundin, A.M., Fuel cells: State-of-the-art and major scientific and engineering problems, Russ. J. Electrochem., 2003, vol. 39, p. 919.
Cornaglia, L.M. and Lombardo, E.A., Pure hydrogen production for low temperature fuel cells, Catal. Lett., 2018, vol.148, p.1015.
Arkhangel’skii, I.V., Dobrovol’skii, Yu.A., Smirnova, T.N., Lyskov, N.V., Dunaev, A.V., Rogacheva, A.E., and Avdeev, V.V., Nizkotemperaturnye toplivnye elementy s protonprovodyashchei polimernoi membranoi: teoreticheskie osnoly, materialy i konstruktsii (Low-temperature Fuel Cells with Proton-exchange Polymer Membrane: Theoretical Fundamentals, Materials, and Structures, Moscow: Moscow State University, 2007).
Sources for Hydrogen. International Energy Agency. Technology Roadmap – Hydrogen and Fuel Cells, Hoeven, M. van der, Ed., Paris: OESC-IEA, 2015.
Trasatti, S., Electrochemical theory. Hydrogen evolution, in Reference Module in Chemistry, Molecular Sciences and Chemical Engineering. Encyclopedia of Electrochemical Power Sources, Reedijk, J., Ed., Amsterdam: Elsevier Science, 2009, p. 41.
Jeremiasse, A., Bergsma, J., Kleijn, M., Saakes, M., Buisman, C., Stuart, M.C., and Hamelers, H.V.M., Performance of metal alloys as hydrogen evolution reaction catalysts in a microbial electrolysis cell, Int. J. Hydrogen. Energy, 2011, vol.36, p.10482.
Kuznetsov, V.V., Gamburg, Yu. D., Zhulikov, V.V., Krutskikh, V.M., Filatova, E.A., Trigub, A.L., and Belyakova, O.A., Electrodeposited NiMo, CoMo, ReNi, and electroless NiReP alloys as cathode materials for hydrogen evolution reaction, Electrochim. Acta, 2020, vol. 354, article no. 136610.
Feng, L., Vrubel, H., Bensimon, M., and Hu, X., Easily-prepared dinickel phosphide (Ni2P) nanoparticles as an efficient and robust electrocatalyst for hydrogen evolution, Phys. Chem. Chem. Phys., 2014, vol. 16, p. 5917.
Lva, Y. and Wang, X., Nonprecious metal phosphides as catalysts for hydrogen evolution, oxygen reduction and evolution reactions, Catal: Sci. Technol., 2017, vol. 7, p.3676.
Benck, J.D., Hellstern, Th.R, Kibsgaard, J., Chakthranont, P., and Jaramillo, Th.F., Catalyzing the hydrogen evolution reaction (HER) with molybdenum sulfide nanomaterials, ACS Catal., 2014, vol. 4, p. 3957.
Heift, D., Iron sulfide materials, catalysts for electrochemical hydrogen evolution, Inorganics, 2019, vol. 7, p. 75.
Tsirlina, G.A. and Petrii, O.A., Hydrogen evolution on smooth stoichiometric tungsten and chromium carbides, Electrochim. Acta, 1987, vol. 32, p. 649.
Zhang, H., Yang, X., Zhang, H., Ma, J., Huang, Zh., Li, J., and Wang, Y., Transition-metal carbides as hydrogen evolution reduction electrocatalysts: Synthetic methods and optimization strategies, Chem.Eur. J., 2021, vol. 27, p. 5074.
Lee, E. and Fokwa, B.P.T., Nonprecious metal borides: Emerging electrocatalysts for hydrogen production, Acc. Chem. Res., 2022, vol. 55, p. 56.
Devadas, B., Hydrogen evolution reaction efficiency by low loading of platinum nanoparticles protected by dendrimers on carbon materials, Electrochem. Commun., 2016, vol. 72, p. 135.
Ji, Zh., Perez-Page, M., Chen J., Rodriguez, R.G., Cai, R., Haigh, S.J., and Holmes, S.M., A structured catalyst support combining electrochemically exfoliated graphene oxide and carbon black for enhanced performance and durability in low-temperature hydrogen fuel cells, Energy, 2021, vol. 226, article no. 120318.
Michalsky, R., Zhang, Y.-L., and Peterson, A.A., Trends in the hydrogen evolution. Activity of metal carbide catalysts, ACS Catal., 2014, vol. 4, p. 1274.
Zhang, H., Yang, X., Zhang, H., Ma, J., Huang, Zh., Li, J., and Wang, Y., Transition-metal carbides as hydrogen evolution reaction electrocatalysts: synthetic methods and optimization strategies, Chem. Eur. J., 2021, vol. 27, p. 5074.
Gao, Q., Zhang, W., Shi, Zh., Yang, L., and Tang, Y., Structural design and electronic modulation of transition-metal-carbide electrocatalysts toward efficient hydrogen evolution, Adv. Mater., 2019, vol. 31, article no. 1802880.
Li, Y., Yin, Z., Liu, X., Cui, M., Chen, S., and Ma, T., Current progress of molybdenum carbide-based materials for electrocatalysis: potential electrocatalysts with diverse applications, Mater. Today Chem., 2021, vol. 19, article no. 100411.
Bayati, M., Liu, X., Abellan, P., Pocock, D., Dixon, M., and Scott, K., Synergistic coupling of a molybdenum carbide nanosphere with Pt nanoparticles for enhanced ammonia electro-oxidation activity in alkaline media, Appl. Energy Mater., 2020, vol. 3, p. 843.
Chen, M., Ma, Y., Zhou, Y., Liu, Ch., Qin, Y., Fang, Y., Guan, G., Li, X., Zhang, Zh., and Wang, T., Influence of transition metal on the hydrogen evolution reaction over nano-molybdenum-carbide catalyst, Catalysts, 2018, vol. 8, p. 294.
Liu, Z., Li, J., Xue, Sh., Zhou, Sh., Qu, K., Li, Y., and Cai, W., Pt/Mo2C heteronanosheets for superior hydrogen evolution reaction, J. Energy Chem., 2020, vol. 47, p. 317.
Podlovchenko, B.I., Gladysheva, T.D., Filatov, A.Yu, and Yashina, L.V., Peculiarities of the Pt(Cu)/C catalyst formation by galvanic displacement of copper in H2PtCl4, Russ. J. Electrochem., 2012, vol. 48, p. 173.]
Podlovchenko, B.I., Krivchenko, V.A., Maksimov, Yu.M., Gladysheva, T.D., Yashina, L.V., Evlashin, S.A., and Pilevsky, A.A., Specific features of the formation of Pt(Cu) catalysts by galvanic displacement with carbon nanowalls used as support, Electrochim. Acta, 2012, vol. 76, p. 137.
Allison, C. and Modin, F.A., Optical and electrical properties of niobium carbide, Phys. Rev. B, 1987, vol. 35, p. 2573.
Ihsan, M., Wang, H., Majid, S., Yang, J., Kennedy, Sh.J., Guo, Z., and Liu, H.K., MoO2/Mo2C/C spheres as anode materials for lithium ion batteries, Carbon, 2016, vol. 96, p. 1200.
Kuznetsov, V.V., Podlovchenko, B.I., Batalov, R.S., and Filatova, E.A., nRu ⋅ mPt ⋅ (Hx – 3n – 2mMoO3) composite prepared by surface redox reaction as a highly active electrocatalyst for carbon monoxide and methanol oxidation, Electrochim. Acta, 2019, vol. 300, p. 274.
Podlovchenko, B.I., Kuznetsov, V.V., and Batalov, R.S., Palladium catalyst modified with molybdenum bronze as a possible alternative to platinum in the methanol oxidation reaction, J. Solid State Electrochem., 2016, vol. 20, p.589.
Kuznetsov, V.V., Batalov, R.S., and Podlovchenko, B.I., nPd0(Hx – 2nMoO3) composites as catalysts of methanol and formic acid electrooxidation, Russ. J. Electrochem., 2016, vol. 52, p. 408.
Hall, M.D., Daly, H.L., Zhang, J.Z., Zhang, M., Alderden, R.A., Pursche, D., Foranc, G.J., and Hambley, T.W., Quantitative measurement of the reduction of platinum(IV) complexes using X-ray absorption near-edge spectroscopy (XANES), Metallomics, 2012, vol. 4, p. 568.
Handbook of Preparative Inorganic Synthesis, Brauer, G., Ed., New York: Academic, 1963.
Trasatti, S. and Petrii, O.A., Real surface area measurements in electrochemistry, Pure Appl. Chem., 1991, vol. 63, p. 711.
Łukaszewski, M., Soszko, M., and Czerwiński, A., Electrochemical methods of real surface area determination of noble metal electrodes – an overview, Int. J. Electrochem. Sci., 2016, vol. 11, p. 4442.
Wyvratt, B.M., Gaudet, J.R., and Thompson, L.T., Effects of passivation on synthesis, structure and composition of molybdenum carbide supported platinum water–gas shift catalysts, J. Catal., 2015, vol. 330, p. 280.
Li, J., Wang, H., and Xiao, X., Intercalation in two-dimensional transition metal carbides and nitrides (MXenes) toward electrochemical capacitor and beyond, Energy Environ. Mater., 2020, vol. 3, p.306.
Bard, A.J., Parsons, R., and Jordan, J., Standard Potentials in Aqueous Solution, New York: M. Dekker, 1985.
Termicheskie konstanty veshchestv. Spravochnik. Chast’ 1. Tablitsy prinyatykh znachenii (Thermal Constants of Substances, Handbook. Part 1. Tables of Accepted Values), Glushko, V.P., Ed., Moscow: VINITI, 1974.
Greczynski, G., Primetzhofer, D., and Hultman, L., Reference binding energies of transition metal carbides by core-level X-ray photoelectron spectroscopy free from Ar+ etching artefacts, Appl. Surf. Sci., 2018, vol. 436, p. 102.
Peiffert, C., Nguyen-Trung, C., Palmer, D.A., Laval, J.P., and Giffaut, E., Solubility of β-Nb2O5 and the hydrolysis of niobium(V) in aqueous solution as a function of temperature and ionic strength, J. Solution Chem., 2010, vol. 39, p. 197.
Kobayashi, T., Sasaki, T., Takagi, I., and Moriyama, H., Solubility of zirconium(IV) hydrous oxides, J. Nucl. Sci. Technol., 2007, vol. 44, p. 90.
Davodi, F., Cilpa-Karhu, G., Sainio, J., Tavakkoli, M., Jiang, H., Mühlhausen, E., Marzun, G., Gökce, B., Laasonen, K., and Kallio, T., Designing of low Pt electrocatalyst through immobilization on metal@C support for efficient hydrogen evolution reaction in acidic media, J. Electroanal. Chem., 2021, vol. 896, article no. 115076.
Chen, W.-F., Iyer, Sh., Iyer, Shw., Sasaki, K., Wang, Ch.-H., Zhu, Y., Muckerman, J.T., and Fujita, E., Biomass-derived electrocatalytic composites for hydrogen evolution, Energy Environ. Sci., 2013, vol. 6, p. 1818.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
Additional information
Translated by T. Safonova
A tribute to outstanding electrochemist Oleg Aleksandrovich Petrii (1937–2021).
Rights and permissions
About this article
Cite this article
Kuznetsov, V.V., Podlovchenko, B.I., Frolov, K.V. et al. The Use of Galvanic Displacement for Synthesizing Pt/Carbide (Mo2C, ZrC, NbC) Catalysts Highly Active in the Hydrogen Evolution Reaction. Russ J Electrochem 58, 896–906 (2022). https://doi.org/10.1134/S1023193522100093
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193522100093