Abstract
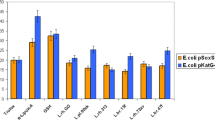
The antigenotoxic and antioxidant activities of biologically active substances of extracts from Inula britannica L. and Limonium gmelinii (Willd.) Kuntze in E. coli strains MG1655 (pColD-lux), MG1655 (pSoxS-lux), and MG1655 (pKatG-lux) were studied by the bioluminescent test. Plant extracts from I. britannica and L. gmelinii in all used concentrations (0.5, 5.0, 50.0, and 500.0 μg/mL) had no genotoxic or oxidant activity. The extracts statistically significantly reduced the bioluminescence intensity of the pColD-lux, pKatG-lux, and pSoxS-lux sensors (p < 0.05) induced by 4-NQO and dioxidine, hydrogen peroxide, and paraquat, respectively. The activity of the extracts depended on their concentration; the greatest antigenotoxic and antioxidant effects were detected at a concentration of 500.0 μg/mL.
Similar content being viewed by others
References
Abilev, S.K. and Glazer, V.M., Mutagenez s osnovami genotoksikologii: uchebnoe posobie (Mutagenesis with the Basics of Genotoxicology: A Tutorial), Moscow: Nestor-Istoriya, 2015.
Natarajan, A., Molnar, P., Sieverdes, K., et al., Microelectrode array recordings of cardiac action potentials as a high throughput method to evaluate pesticide toxicity, Toxicol. In Vitro, 2006, vol. 20, no. 3, pp. 375–381. doi 10.1016/j.tiv.2005.08.014
Holland, N.T., Duramad, P., Rothman, N., et al., Micronucleus frequency and proliferation in human lymphocytes after exposure to herbicide 2,4-dichlorophenoxyacetic acid in vitro and in vivo, Mutat. Res. Gen. Toxicol. Environ. Mutagen., 2002, vol. 521, nos. 1–2, pp. 165–178. doi 10.1016/S1383-5718(02)00237-1
Goncharova, R.I. and Kuzhir, T.D., Molecular basis of applying antimutagens as anticarcinogens, Ekol. Genet., 2005, vol. 3, no. 3, pp. 19–32.
Durnev, A.D., Methodical aspects of research on modification of chemical mutagenesis, Byull. Eksp. Biol. Med., 2008, vol. 146, no. 9, pp. 281–287. doi 10.1007/s10517-008-0273-5
Uzun, F., Kalender, S., Durak, D., et al., Malathioninduced testicular toxicity in male rats and the protective effect of vitamins C and E, Food Chem. Toxicol., 2009, vol. 47, no. 8, pp. 1903–1908. doi 10.1016/j.fct.2009.05.001
Słoczyńska, K., Powroźnik, B., Pękala, E., and Waszkielewicz, A.M., Antimutagenic compounds and their possible mechanisms of action, J. Appl. Genet., 2014, vol. 55, no. 2, pp. 273–285. doi 10.1007/s13353-014-0198-9
Seca, A.M.L., Grigore, A., Pinto, D.C.C.A., and Silva, A.M.S., The genus Inula and their metabolites: from ethnopharmacological to medicinal uses, J. Ethnopharmacol., 2014, vol. 154, no. 2, pp. 286–310. doi 10.1016/j.jep.2014.04.010
Kolumbaeva, S.Zh., Lovinskaya, A.V., Zhusupova, A.I., et al., Toxic and mutagenic activity of biologically active substances from the Limonium gmelinii plants, family Plumbaginaceae (=Limoniaceae Lincz.), Vestn. Kaz. Natc. Univ., Ser. Biol., 2016, vol. 66, no. 1, pp. 144–153.
Kolumbaeva, S.Zh., Lovinskaya, A.V., Akhtaeva, N.Z., et al., Toxic and mutagenic activity of biologically active substances from the Inula britannica L. plants, family Compositae, Vestn. Kaz. Natc. Univ., Ser. Biol., 2016, vol. 69, no. 4, pp. 134–145.
Manukhov, I.V., Kotova, V.Yu., Mal’dov, D.G., et al., Induction of oxidative stress and SOS response in Escherichia coli by vegetable extracts: the role of hydroperoxides and the synergistic effect of simultaneous treatment with cisplatinum, Microbiology (Moscow), 2008, vol. 77, pp. 523–529. https://doi.org/10.1134/S0026261708050020.
Kotova, V.Yu., Manukhov, I.V., and Zavigel’skii, G.B., Lux-biosensors for the detection of SOS-response, heat shock and oxidative stress, Biotekhnologiya, 2009, no. 6, pp. 16–25. doi 10.1134/S0003683810080089
Zavilgelsky, G.B., Kotova, V.Y., and Manukhov, I.V., Action of 1,1-dimethylhydrazine on bacterial cells is determined by hydrogen peroxide, Mutat. Res., 2007, vol. 634, nos. 1–2, pp. 172–176. doi 10.1016/j.mrgentox.2007.07.012
Maniatis, T., Fritsch, E.F., and Sambrook, J., Molecular Cloning: A Laboratory Manual, Cold Spring Harbor: Cold Spring Harbor Lab., 1982.
Farr, S.B. and Kogoma, T., Oxidative stress responses in Escherichia coli and Salmonella typhimurium, Microbiol. Rev., 1991, vol. 55, pp. 561–585.
Downes, D.J., Chonofsky, M., Tan, K., et al., Characterization of the mutagenic spectrum of 4-nitroquinoline 1-oxide (4-NQO) in Aspergillus nidulans by whole genome sequencing, G3 (Bethesda), 2014, vol. 4, no. 12, pp. 2483–2492. doi 10.1534/g3.114.014712
Padeiskaya, E.N., Antibacterial preparation Dioxydin: biological effect and importance in the therapy of various forms of purulent infection, Infekts. Antimikrobn. Ter., 2001, no. 5, pp. 150–155.
Sycheva, L.P., Kovalenko, M.A., Sheremet’eva, S.M., et al., Study of the mutagenic action of dioxidine by a multi-organ micronucleus method, Byull. Eksp. Biol. Med., 2004, vol. 138, no. 8, pp. 188–190.
Ordzhonikidze, K.G., Zanadvorova, A.M., and Abilev, S.K., Organ specificity of the genotoxic effects of cyclophosphane and dioxidine: an alkaline comet assay study, Russ. J. Genet., 2011, vol. 47, no. 6, pp. 754–756.
Rzezniczak, T.Z., Douglas, L.A., Watterson, J.H., et al., Paraquat administration in Drosophila for use in metabolic studies of oxidative stress, Anal. Biochem., 2011, vol. 419, no. 2, pp. 345–347. doi 10.1016/j.ab.2011.08.023
Havsteen, B.H., The biochemistry and medical significance of the flavonoids, Pharmacol. Ther., 2002, vol. 96, nos. 2–3, pp. 67–202. doi 10.1016/S0163-7258(02)00298-X
Lin, Y., Shi, R., Wang, X., et al., Luteolin, a flavonoid with potential for cancer prevention and therapy, Curr. Cancer Drug Targets, 2008, vol. 8, no. 2, pp. 634–646. doi 10.2174/156800908786241050
Skopichev, V.G., Bogolyubova, I.O., Zhichkina, L.V., and Maksimyuk, N.N., Ekologicheskaya fiziologiya (Ecological Physiology), St. Petersburg: Kvadro, 2014.
De Flora, S., Mechanisms of inhibitors of mutagenesis and carcinogenesis, Mutat. Res., 1998, vol. 402, pp. 151–158.
Bouhlel, I., Mansour, H.B., Limem, I., et al., Screening of antimutagenicity via antioxidant activity in different extracts from the leaves of Acacia salicina from the center of Tunisia, Environ. Toxicol. Pharmacol., 2007, vol. 23, no. 1, pp. 56–63. doi 10.1016/j.etap.2006.07.001
Wu, Ch.-H. and Yen, G.-Ch., Antigenotoxic properties of Cassia tea (Cassia tora L.): mechanism of action and the influence of roasting process, Life Sci., 2004, vol. 76, no. 1, pp. 85–101. doi 10.1016/j.lfs.2004.07.011
Tsao, R. and Deng, Z., Separation procedures for naturally occurring antioxidant phytochemicals, J. Chromatogr., 2004, vol. 812, pp. 85–99. doi 10.1016/j.jchromb.2004.09.028
Igonina, E.V., Marsova, M.V., and Abilev, S.K., Luxbiosensors: screening of biologically active compounds for genotoxicity, Ekol. Genet., 2016, vol. 14, no. 4, pp. 52–62. doi 10.17816/ecogen14452-14462
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.V. Lovinskaya, S.Zh. Kolumbayeva, T.M. Shalakhmetova, M.V. Marsova, S.K. Abilev, 2017, published in Genetika, 2017, Vol. 53, No. 12, pp. 1393–1401.
Rights and permissions
About this article
Cite this article
Lovinskaya, A.V., Kolumbayeva, S.Z., Shalakhmetova, T.M. et al. Antigenotoxic activity of biologically active substances from Inula britannica and Limonium gmelini . Russ J Genet 53, 1311–1319 (2017). https://doi.org/10.1134/S1022795417120080
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1022795417120080