Abstract
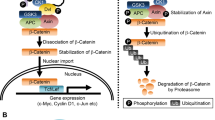
The protein Merlin is involved in the regulation of cell proliferation and differentiation in the eyes and wings of Drosophila and is a homolog of the human protein encoded by the Neurofibromatosis 2 (NF2) gene whose mutations cause auricular nerve tumors. Recent studies show that Merlin and Expanded cooperatively regulate the recycling of membrane receptors, such as the epidermal growth factor receptor (EGFR). By performing a search for potential genetic interactions between Merlin (Mer) and the genes important for vesicular trafficking, we found that ectopic expression in the wing pouch of the clathrin adapter protein Lap involved in clathrin-mediated receptor endocytosis resulted in the formation of extra vein materials. On the one hand, coexpression of wild-type Merlin and lap in the wing pouch restored normal venation, while overexpression of a dominant-negative mutant Mer DBB together with lap enhanced ectopic vein formation. Using various constructs with Merlin truncated copies, we showed the C-terminal portion of the Merlin protein to be responsible for the Merlin-lap genetic interaction. Furthermore, we showed that the Merlin and Lap proteins colocalized at the cortex of the wing imaginal disc cells.
Similar content being viewed by others
References
Held, L.I., Imaginal Discs: The Genetic and Cellular Logic of Pattern Formation, Cambridge: Cambridge Univ. Press, 2002.
Blair, S.S., Wing Vein Patterning in Drosophila and the Analysis of Intercellular Signaling, Ann. Rev. Cell Dev. Biol., 2007, vol. 23, pp. 293–319.
LaJeunesse, D.R., McCartney, B.M., and Fehon, R.G., Structural Analysis of Drosophila Merlin Reveals Functional Domains Important for Growth Control and Subcellular Localization, J. Cell Biol., 1998, vol. 141, pp. 1589–1599.
Maitra, S., Kulikauskas, R.M., Gavilan, H., and Fehon, R.G., The Tumor Suppressors Merlin and Expanded Function Cooperatively to Modulate Receptor Endocytosis and Signaling, Curr. Biol., 2006, vol. 16, pp. 702–709.
Rouleau, G.A., Merel, P., Luchtman, M., et al., Alteration in a New Gene Encoding a Putative Membrane-Organizing Protein Causes Neurofibromatosis Type 2, Nature, 1993, vol. 363, pp. 515–521.
Trofatter, J.A., MacCollin, M.M., Rutter, J.L., et al., A Novel Moesin-, Exrin-, Radixin-like Gene Is a Candidate for the Neurofibromatosis 2 Tumor-Suppressor, Cell, 1993, vol. 72, pp. 791–800.
Algrain, M., Arpin, M., and Louvard, D., Wizardry at the Cell Cortex, Curr. Biol., 1993, vol. 3, pp. 451–454.
Golovnina, K., Blinov, A., Akhmametyeva, E.M., et al., Evolution and Origin of Merlin, the Product of the Neurofibromatosis Type 2 (NF2) Tumor-Suppressor Gene, BMC Evol. Biol., 2005, vol. 5, pp. 69–86.
LaJeunesse, D.R., McCartney, B.M., and Fehon, R.G., A Systematic Screen for Dominant Second-Site Modifiers of Merlin/NF2 Phenotypes Reveals an Interaction with Blistered/DSRF and Scribbler, Genetics, 2001, vol. 158, pp. 667–679.
Seto, E.S., Bellen, H.J., and Lloyd, T.E., When Cell Biology Meets Development: Endocytic Regulation of Signaling Pathways, Genes Dev., 2002, vol. 16, pp. 1314–1336.
Kirchhausen, T., Bonifacino, J.S., and Riezman, H., Linking Cargo to Vesicle Formation: Receptor Tail Interactions with Coat Proteins, Curr. Opin. Cell. Biol., 1997, vol. 9, pp. 488–495.
Zhang, B., Koh, Y.H., Beckstead, R.B., et al., Synaptic Vesicle Size and Number Are Regulated by a Clathrin Adaptor Protein Required for Endocytosis, Neuron, 1998, vol. 21, pp. 1465–1475.
Zhang, B., Ganetzky, B., Bellen, H.J., and Murthy, V.N., Tailoring Uniform Coats for Synaptic Vesicles during Endocytosis, Neuron, 1999, vol. 23, pp. 419–422.
Wang, L.H., Sudhof, T.C., and Anderson, R.G., The Appendage Domain of Alpha-Adaptin Is a High Affinity Binding Site for Dynamin, J. Biol. Chem., 1995, vol. 270, pp. 10079–10083.
Ringstad, N., Nemoto, Y., and De Camilli, P., The SH3p4/Sh3p8/SH3p13 Protein Family: Binding Partners for Synaptojanin and Dynamin via a Grb2-Like Src Homology 3 Domain, Proc. Natl. Acad. Sci. USA, 1997, vol. 94, no. 15, pp. 8569–8574.
Newmyer, S.L. and Schmid, S.L., Dominant-Interfering Hsc70 Mutants Disrupt Multiple Stages of the Clathrin-Coated Vesicle Cycle in vivo, J. Cell Biol., 2001, vol. 152, pp. 607–620.
Griffiths, G. and Gruenberg, J., The Arguments for Preexisting Early and Late Endosomes, Trends Cell Biol., 1991, vol. 1, no. 1, pp. 5–9.
Murphy, R.F., Maturation Models for Endosome and Lysosome Biogenesis, Trends Cell Biol., 1991, vol. 1, pp. 77–82.
Seaman, M.N.J. and Luzio, J.P., Lysosomes and Other Late Compartments of the Endocytic Pathway, in Endocytosis: Frontiers in Molecular Biology, Oxford: Oxford Univ. Press, 2001, pp. 111–148.
Selva, E.M., Hong, K., Baeg, G.H., et al., Dual Role of the Fringe Connection Gene in Both Heparan Sulphate and Fringedependent Signalling Events, Nat. Cell Biol., 2001, vol. 3, pp. 809–815.
Entchev, E.V., Schwabedissen, A., and GonzalezGaitan, M., Gradient Formation of the TGF-Beta Homolog Dpp, Cell, 2000, vol. 103, pp. 981–991.
Tanaka, K., Kitagawa, Y., and Kadowaki, T., Drosophila Segment Polarity Gene Product Porcupine Stimulates the Posttranslational N-Glycosylation of Wingless in the Endoplasmic Reticulum, J. Biol. Chem., 2002, vol. 277, pp. 12816–12823.
Capdevila, J. and Guerrero, I., Targeted Expression of the Signaling Molecule Decapentaplegic Induces Pattern Duplications and Growth Alterations in Drosophila Wings, EMBO J., 1994, vol. 13, pp. 4459–4468.
Coelho, C.M. and Leevers, S.J., Do Growth and Cell Division Rates Determine Cell Size in Multicellular Organisms?, J. Cell Sci., 2000, vol. 113, pp. 2927–2934.
McCartney, B.M. and Fehon, R.G., Distinct Cellular and Subcellular Patterns of Expression Imply Distinct Functions for the Drosophila Homologues of Moesin and the Neurofibromatosis 2 Tumor Suppressor, merlin, J. Cell Biol., 1996, vol. 133, pp. 843–852.
Kirchhausen, T., Clathrin, Annu. Rev. Biochem., 2000, vol. 69, pp. 699–727.
Slepnev, V.I. and De Camilli, P., Accessory Factors in Clathrin-Dependent Synaptic Vesicle Endocytosis, Nat. Rev. Neurosci., 2000, vol. 1, pp. 161–172.
Lafer, E.M., Clathrin-Protein Interactions, Traffic, 2002, vol. 3, pp. 513–520.
Zhou, S., Sousa, R., Tannery, N.H., and Lafer, E.M., Characterization of a Novel Synapsespecific Protein: II. cDNA Cloning and Sequence Analysis of the F1-20 Protein, J. Neurosci., 1992, vol. 12, pp. 2144–2155.
Schroder, S., Morris, S.A., Knorr, R., et al., Primary Structure of the Neuronal Clathrin-Associated Protein Auxilin and Its Expression in Bacteria, Eur. J. Biochem., 1995, vol. 228, pp. 297–304.
Curto, M., Cole, B.K., Lallemand, D., et al., ContactDependent Inhibition of EGFR Signaling by Nf2/Merlin, J. Cell Biol., 2007, vol. 177, pp. 893–903.
Gutmann, D.H., Sherman, L., Seftor, L., et al., Increased Expression of the NF2 Tumor Suppressor Gene Product, Merlin, Impairs Cell Motility, Adhesion and Spreading, Hum. Mol. Genet., 1999, vol. 8, pp. 267–275.
Zhai, L., Chaturvedi, D., and Cumberledge, S., Drosophila Wnt-1 Undergoes a Hydrophobic Modification and Is Targeted to Lipid Rafts, a Process That Requires Porcupine, J. Biol. Chem., 2004, vol. 279, pp. 33220–33227.
Nusse, R., Wnts and Hedgehogs: Lipid-Modified Proteins and Similarities in Signaling Mechanisms at the Cell Surface, Development, 2003, vol. 130, pp. 5297–5305.
Johnston, L.A. and Edgar, B.A., Wingless and Notch Regulate Cell-Cycle Arrest in the Developing Drosophila Wing, Nature, 1998, vol. 394, pp. 82–84.
Kopyl, S.A., Dorogova, N.V., Baimak, T.Yu., et al., Role of the porcupine Gene in the Development of the Wing Imaginal Disk of Drosophila melanogaster, Russ. J. Genet., 2008, vol. 44, no. 11, pp. 1290–1295.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © S.A. Kopyl, N.V. Dorogova, E.M. Akhmametyeva, L.V. Omelyanchuk, L.-S. Chang, 2010, published in Genetika, 2010, Vol. 46, No. 3, pp. 314–320.
Rights and permissions
About this article
Cite this article
Kopyl, S.A., Dorogova, N.V., Akhmametyeva, E.M. et al. Drosophila melanogaster gene Merlin interacts with the clathrin adaptor protein gene lap. Russ J Genet 46, 276–282 (2010). https://doi.org/10.1134/S1022795410030038
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1022795410030038