Abstract
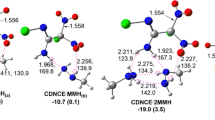
The quantum-chemical simulation of the interactions underlying the absorption extraction process of hydrogen sulfide from gas streams with diethanolamine (DEA) has been performed. It has been shown that molecular complexes are formed without any barrier in the gas phase, whereas both molecular and ionic complexes are formed in the aqueous phase. It has been found that the use of the IPCM continuum solvation model fails to adequately reproduce the value of the heat of the reaction. A promising method for describing the absorption process of hydrogen sulfide extraction with alkanolamines using the supramolecular approach based on a discrete solvation model has been proposed. The approach takes account of intermolecular interactions in the DEA–H2S–H2O system and makes it possible to determine structural and energy parameters of the resulting associates and the mechanism of the process.
Similar content being viewed by others
References
V. I. Murin, N. N. Kislenko, and Yu. V. Surkov, Natural Gas and Condensate Processing Technology (Nedra, Moscow, 2002) [in Russian].
A. Yu. Adzhiev and P. A. Purtov, Conditioning and Processing of Associated Petroleum Gas in Russia (EDVI, Krasnodar, 2014), part 1 [in Russian].
T. A. Semenova, I. L. Leites, and Yu. V. Aksel’rod, Purification of Process Gases (Khimiya, Moscow, 1977) [in Russian].
Handbook of Modern Gas Processing Processes: Oil, Gas, and Petroleum Chemistry Abroad, No. 4.65 (1984).
T. G. Alkhazov and N. S. Amirgulyan, Sulfur Compounds in Natural Gases and Crude Oils (Nedra, Moscow, 1989) [in Russian].
L. I. Borshchenko, Preparation of Gas and Condensate for Transport (Nedra, Moscow, 1987) [in Russian].
L. Kohl and R. Nielsen, Gulf Profess. Publ., No. 5, 900 (1997).
J. L. de Medeiros, L. C. Barbosa, O. de Queiroz, and F. Araujo, Ind. Eng. Chem. Res. 52, 9203 (2013).
Kirk-Othmer Encyclopedia of Chemical Technology (Wiley, New York, 2004), 5th Ed., Vol. 7, p. 91.
T. R. Prosochkina, E. L. Artem’eva, and E. A. Kantor, Russ. J. Gen. Chem. 83, 10 (2013).
M. S. Tokar’, E. L. Artem’eva, T. R. Prosochkina, and E. A. Kantor, Bashkir. Khim. Zh. 10, 81 (2003).
A. A. Granovsky. http://classic.chem.msu.su/gran/gamess/index.html.
A. D. Becke, J. Chem. Phys. 98, 5648 (1993).
C. Lee, W. Yang, and R. G. Parr, Phys. Rev. 37, 785 (1988).
C. Möller and M. S. Plesset, Phys. Rev. 46, 618 (1934).
J. B. Foresman, T. A. Keith, K. B. Wiberg, et al., J. Phys. Chem. 100, 16098 (1996).
A. P. Scott and L. Radom, J. Phys. Chem. 100, 16502 (1996).
H. G. Chang, Nat. Gas Ind. 15 (6), 61 (1995).
M. I. Shakhparonov, Introduction to the Molecular Theory of Solutions (Gos. Izd. Tekhniko-Teoreticheskoi Literatury, Moscow, 1956) [in Russian].
B. Ya. Simkin and I. I. Sheikhet, Quantum-Chemical and Statistical Theory of Solutions: Computational Methods and Their Application (Khimiya, Moscow, 1989) [in Russian].
J. H. Lii, B. Ma, and N. L. Allinger, J. Chem. 20, 1593 (1999).
S. Li, F. Tao, and R. Gu, Chem. Phys. Lett. 417, 434 (2006).
S. S. Konyukhov, A. A. Moskovskii, D. A. Firsov, and A. V. Nemukhin, Vestn. Mosk. Univ., Ser. 2: Khim. 43, 4 (2002).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © T.R. Prosochkina, A.P. Nikitina, E.A. Kantor, 2016, published in Neftekhimiya, 2016, Vol. 56, No. 4, pp. 384–391.
Rights and permissions
About this article
Cite this article
Prosochkina, T.R., Nikitina, A.P. & Kantor, E.A. Hydrogen sulfide removal from hydrocarbon gas mixtures by diethanolamine (computer simulation). Pet. Chem. 56, 616–622 (2016). https://doi.org/10.1134/S0965544116070136
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0965544116070136