Abstract
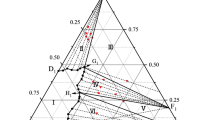
Phase equilibria are studied in ternary contouring systems and in the CO(NH2)2-KH2PO4-K2HPO4-H2O quaternary system at 25°C using an optimized method of cross sections. It is found that an incongruent chemical compound forms between the components of a system with the composition 2KH2PO4 · K2HPO4. The compositions of eutonic and peritonic solutions and the solid phases saturating them are determined. The lines of monovariant equilibria and the crystallization surface of potassium dihydrogen phosphate are studied. The location of the eutonic and peritonic solutions and monovariant equilibrium lines near the plane are shown.
Similar content being viewed by others
References
M. Foroutan, J. Chem. Eng. Data 52, 859 (2007).
D. Kalina and P. Kirilov, J. Univ. Chem. Technol. Metallurg., Sofia, Bulgaria 55, 2225 (2010).
K. Kamburova, P. Kirilov, and L. Brakalov, J. Univ. Chem. Technol. Metallurg., Sofia, Bulgaria 44, 249 (2009).
M. Kabiri-Badr and M. T. Zafarani-Moatter, J. Univ. Chem. Technol. Metallurg., Sofia, Bulgaria 40, 412 (1995).
S. A. Mazunin, Physicochemical Analysis in Chemistry and Chemical Technology (Perm. Gos. Univ., Perm, 2014) [in Russian].
N. S. Kistanova, Extended Abstract of Candidate’s Dissertation in Chemistry (Perm. State Natl. Res. Univ., Perm, 2011).
N. S. Kistanova, S. A. Mazunin, S. I. Frolova, et al., Vestn. Tambov. Gos. Tekh. Univ. 16, 863 (2010).
S. A. Mazunin, S. I. Frolova, and N. S. Kistanova, RF Patent No. 2421721, Byull. Izobret. No. 17 (2011).
V. L. Chechulin and S. A. Mazunin, Izv. Vyssh. Uchebn. Zaved., Khim. Tekhnol. 53(3), 152 (2010).
V. L. Chechulin and S. A. Mazunin, Russ. J. Gen. Chem. 82, 199 (2012).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © M.N. Noskov, S.A. Mazunin, 2015, published in Zhurnal Fizicheskoi Khimii, 2015, Vol. 89, No. 6, pp. 971–977.
Rights and permissions
About this article
Cite this article
Noskov, M.N., Mazunin, S.A. Study of phase equilibria in the CO(NH2)2-KH2PO4-K2HPO4-H2O system at 25°C using an optimized method of cross sections. Russ. J. Phys. Chem. 89, 1011–1017 (2015). https://doi.org/10.1134/S0036024415060217
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024415060217