Abstract
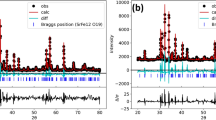
Single crystals of lithium molybdate tungstates containing 10 and 15 mol % molybdenum were grown by the low-temperature-gradient Czochralski technique. It was shown that the single crystals have the phenakite structure (space group \(R\bar {3}\)). The heat capacity of the Li2W0.85Mo0.15O4 single crystal was measured by differential scanning calorimetry in two temperature ranges: 190–370 and 320–970 K. The heat capacity in the temperature range 190–970 K is well described by the equation Cp = 37.95541 + 0.42875T − 4.87201 × 10–4T2 + 2.15184 × 10–7T 3 J/(K mol). It was determined that there are no phase transitions in the Li2W0.85Mo0.15O4 single crystal in the studied temperature range. This makes this single crystal promising for the search for rare events, such as neutrinoless double β-decay and elastic coherent neutrino scattering off nuclei.




Similar content being viewed by others
Change history
20 December 2022
An Erratum to this paper has been published: https://doi.org/10.1134/S0036023622340010
REFERENCES
O. Barinova, A. Sadovskiy, I. Ermochenkov, et al., J. Cryst. Growth 468, 365 (2017). https://doi.org/10.1016/j.jcrysgro.2016.10.009
K. S. Gavrichev, N. N. Smirnova, A. V. Khoroshilov, et al., Russ. J. Inorg. Chem. 52, 727 (2007). https://doi.org/10.1134/S0036023607050129
N. I. Matskevich, V. N. Shlegel, A. L. Sednev, et al., J. Chem. Thermodyn. 143, 106059 (2020). https://doi.org/10.1016/j.jct.2020.106059
T. L. Simonenko, V. A. Bocharova, N. P. Simonenko, et al., Russ. J. Inorg. Chem. 66, 1779 (2021). https://doi.org/10.1134/S0036023621120160
A. Aliane, I. Avetissov, O. Barinova, et al., Nucl. Instrum. Methods Phys. Res., Sect. A 949, 162784 (2020). https://doi.org/10.1016/j.nima.2019.162784
Z. A. Fattakhova, E. G. Vovkotrub, and G. Z. Zakharova, Russ. J. Inorg. Chem. 66, 35 (2021). https://doi.org/10.1134/S0036023621010022
L. M. Kustov, A. L. Kustov, and T. Salmi, Mendeleev Commun. 32, 1 (2022). https://doi.org/10.1016/j.mencom.2022.01.001
F. Xu, G. Zhang, M. Luo, et al., Nat. Sci. Rev. 8, 104 (2021). https://doi.org/10.1093/nsr/nwaa104
A. V. Khoroshilov, G. A. Sharpataya, K. S. Gavrichev, et al., Russ. J. Inorg. Chem. 57, 1123 (2012). https://doi.org/10.1134/S0036023612080074
M. V. Makarova, P. E. Kazin, Yu. D. Tretyakov, et al., Physica C 419, 61 (2005). https://doi.org/10.1016/j.physc.2004.12.003
C. W. F. T. Pistorius, J. Solid State Chem. 13, 325 (1975). https://doi.org/10.1016/0022-4596(75)90147-4
V. V. Atuchin, V. L. Bekenev, Yu. A. Borovlev, et al., J. Opt. Adv. Mater. 19, 86 (2017).
I. R. Pandey, S. Karki, D. J. Daniel, et al., J. Alloys Compd. 860, 158510 (2021). https://doi.org/10.1016/j.jallcom.2020.158510
V. D. Grigorieva, V. N. Shlegel, Y. A. Borovlev, et al., J. Cryst. Growth 523, 125144 (2019). https://doi.org/10.1016/0022-4596(75)90147-4
L. Berge, R. S. Boiko, M. Chapellier, et al., JINST P06004 (2014). https://doi.org/10.1088/1748-0221/9/06/P06004
I. M. Ivanov, Yu. G. Stenin, V. N. Shlegel, et al., Inorg. Mater. 44, 1330 (2008). https://doi.org/10.1134/S0020168508120121
N. I. Matskevich, T. Wolf, M. Le Tacon, et al., J. Therm. Anal. Calorim. 130, 1125 (2017). https://doi.org/10.1007/s10973-017-6493-z
I. A. Uspenskaya, Z. S. Vakhovskaya, M. M. Efremova, et al., Russ. J. Phys. Chem. A 80, 529 (2006). https://doi.org/10.1134/S0036024406040078
D. A. Samoshkin, A. Sh. Agazhanov, and S. V. Stankus, J. Phys.: Conf. Series 2119, 012138 (2021). https://doi.org/10.1088/1742-6596/2119/1/012138
L. Denielou, J. P. Petitet, and C. Tequi, J. Chem. Thermodyn. 7, 901 (1975). https://doi.org/10.1016/0021-9614(75)90100-7
M. Velazquez, P. Veber, and M. Moutatouia, Solid State Sci. 65, 41 (2017). https://doi.org/10.1016/j.solidstatesciences.2016.12.006
A. E. Musikhin, V. N. Naumov, M. A. Bespyatov, et al., J. Alloys Compd. 639, 145 (2015). https://doi.org/10.1016/j.jallcom.2015.03.159
A. E. Musikhin, M. A. Bespyatov, T. M. Kuzin, et al., J. Phys.: Conf. Ser. 2119, 012137 (2021). https://doi.org/10.1088/1742-6596/2119/1/012137
E. Gamsjager and M. Wiessner, Monatsh Chem. 149, 357 (2018). https://doi.org/10.1007/s00706-017-2117-3
V. N. Naumov and A. E. Musikhin, Comput. Mater. Sci. 130, 257 (2017). https://doi.org/10.1016/j.commatsci.2017.01.025
Funding
This work was supported by the Russian Science Foundation (project no. 19-19-00095-P). V. N. Shlegel’, V. D. Grigor’eva, A. N. Semerikova, and V. A. Kuznetsov thank the Ministry of Science and Higher Education of the Russian Federation (project no. 121031700314-5) for using the calorimeter and the setups for growing single crystals.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by V. Glyanchenko
The original online version of this article was revised: Surname of the third author should read Grigorieva.
Rights and permissions
About this article
Cite this article
Matskevich, N.I., Shlegel’, V.N., Grigorieva, V.D. et al. Crystal Growth and Heat Capacity of Lithium Molybdate Tungstates. Russ. J. Inorg. Chem. 67, 1521–1526 (2022). https://doi.org/10.1134/S0036023622600812
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023622600812