Abstract
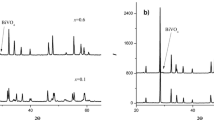
Phase equilibria were studied in the Nb2O5–CdO system in the Nb2O5-rich region including CdNb2O6 and Cd2Nb2O7. It was determined that CdNb2O6 and Cd2Nb2O7 in air are stable to 1150 and 1120°C, respectively, and that, above these temperatures, there is solid-phase decomposition of niobates with CdO release in the gas phase. Along with the cadmium oxide evaporation, the Cd2Nb2O7 decomposition is accompanied by the formation of cadmium metaniobate CdNb2O6 and the CdNb2O6 decomposition results in the formation of niobium oxide Nb2O5. No thermal events were observed in the differential thermal analysis curve for a 1: 1 CdNb2O6–Cd2Nb2O7 mixture heated to 1100°C in air, which suggests that there are neither phase transformations in cadmium niobates, nor a eutectic within this temperature and concentration ranges. A study of the morphology of compacted samples of niobates determined specific conditions for producing dense composite ceramics, a mixture of niobates, that is suitable for using as a dielectric material.
Similar content being viewed by others
References
E. Weiner and C. Wentworth, J. Am. Ceram. Soc. 35, 207 (1952).
F. Jona, G. Shirane, and R. Pepinsky, Phys. Rev. 98, 903 (1955).
V. A. Isupov, Phys. Sol. State 47, 2119 (2005).
N. N. Kolpakova, R. Margraf, and M. Polomska, J. Phys.: Condens. Matter 6, 2787 (1994).
K. Lukaszewicz, A. Pietraszko, J. Stepien-Damm, and N. N. Kolpakova, Mater. Res. Bull., No. 29, 987 (1994).
N. N. Kolpakova, M. Wiesner, A. O. Lebedev, et al., Tech. Phys. Lett. 24, 679 (1998).
C. M. Ronconi and O. L. Alves, Thin Solid Films 441, 121 (2003).
G. A. Samara, E. L. Venturini, and L. A. Boatner, J. Appl. Phys. 100, 074112 (2006).
F. X. Zhang, J. Lian, U. Becker, et al., Phys. Rev. 74B, 174116 (2006).
W. N. Lawless, Ferroelectrics 43, 223 (1982).
C. Ang, R. Guo, A. S. Bhalla, and L. E. Gross, J. Appl. Phys. 87, 7452 (2000).
C. Ang, A. S. Bhalla, R. Guo, and L. E. Gross, J. Appl. Phys. 90, 2465 (2001).
N. N. Kolpakova, J. Exp. Theor. Phys. 96, 538 (2003).
Zhi. Yu and C. Ang, Appl. Phys. Lett. 85 (587), 801 (2004).
Y.-J. Hsiao, G.-C. Chang, and T.-H. Chang, J. Alloys Compd. 471, 259 (2009).
Y.-J. Hsiao, T.-H. Fang, L.-W. Ji, and S.-S. Chi, Open Surf. Sci. J. 1, 30 (2009).
M. K. Ekmekçi, M. Erdem, A. Mergen, et al., J. Alloys Compd. 591, 230 (2014).
L. Reed, J. Am. Ceram. Soc. 44, 576 (1961).
R. S. Roth, J. Am. Ceram. Soc. 44, 49 (1961).
S. L. Swartz, C. A. Randall, and A. S. Bhalla, J. Am. Ceram. Soc. 72, 637 (1989).
S. Waplak and N. N. Kolpakova, Phys. Status Solidi A 117, 461 (1990).
A. de Bretteville, F. A. Halden, T. Valisos, and L. Reed, J. Am. Ceram. Soc. 40, 86 (1957).
Yu. V. Karyakin and I. I. Angelov, Pure Chemical Reagents (Khimiya, Moscow, 1974) [in Russian].
W. R. Cook and H. Jaffe, Phys. Rev. 88, 1426 (1952).
W. R. Cook and H. Jaffe, Phys. Rev. 89, 1297 (1953).
B. H. Toby, J. Appl. Crystallogr. 34, 210 (2001).
A. C. Larson and R. B. Von Dreele, General Structure Analysis System (GSAS) Los Alamos National Laboratory Report LAUR 86 (2004).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © R.F. Samigullina, M.V. Rotermel, I.V. Nikolaenko, T.I. Krasnenko, 2016, published in Zhurnal Neorganicheskoi Khimii, 2016, Vol. 61, No. 2, pp. 167–172.
Rights and permissions
About this article
Cite this article
Samigullina, R.F., Rotermel, M.V., Nikolaenko, I.V. et al. Phase equilibria in the Nb2O5–CdO system and the thermal stability of Cd2Nb2O7 and CdNb2O6 . Russ. J. Inorg. Chem. 61, 156–160 (2016). https://doi.org/10.1134/S0036023616020182
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023616020182