Abstract
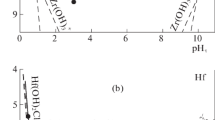
Gibbs energies of formation have been determined by conductometric titration for hydroxo complexes of cerium, samarium, europium, erbium, ytterbium, and yttrium. All these elements form monohydroxo complexes; yttrium, erbium, and terbium also form dihydroxo complexes. The Gibbs energies of formation of lanthanide hydroxo complexes from ions have virtually equal values of −47.3 ± 0.6 kJ/mol monohydroxo complexes and −44.5 ± 0.5 kJ/mol for dihydroxo complexes, respectively. These values were used to estimate the Gibbs energies of formation of hydroxo complexes for the entire lanthanide series.
Similar content being viewed by others
References
V. R. Sastri, J. R. Perumareddi, RaoV. Ramachandra, et al., Modern Aspects of Rare Earth and Their Complexes (Elsevier, Amsterdam, 2003).
A. S. Isaeva, I. I. Kozhina, A. M. Toikka, and I. A. Zvereva, Glass Phys. Chem. 32(1), 106 (2006).
R. B. Grieves and W. R. Charewicz, Sep. Sci. Technol. 10(1), 77 (1975).
B. B. Gassett, J. S. Otis, and F. A. Peter, J. Water Pollution Control Fed. 37(4), 460 (1965).
N. N. Voronin, V. V. Demidov, A. V. Cherkasov, and I. P. Antonova, Zh. Prikl. Khim. 65, 2005 (1992).
D. E. Chirkst, T. E. Litvinova, V. S. Starshinova, and G. S. Roshchin, Zh. Prikl. Khim. 80, 187 (2007).
V. S. Starshinova, Zap. Gornogo Inst. 170(1), 172 (2007).
D. E. Chirkst and O. L. Lobacheva, Zh. Prikl. Khim. 82, 1273 (2009).
www.chem.msu.su. TKV Database. Parameters and definitions.
HSC Chemistry Outo Kumpu Research OY (ver. 4.1.), Pori, Finland.
I. N. Beloglazov and S. Z. El’-Salim, Processing of Experimental Results (Ruda i Metally, 2004) [in Russian].
A. A. Ravdel’ and A. M. Ponomareva, A Concise Reference Book of Physicochemical Quantities (Ivan Fedorov, St. Petersburg, 2003) [in Russian].
F. A. Cotton and G. Wilkinson, Basic Inorganic Chemistry (Wiley, New York, 1976), Vol. 3.
S. A. Shchukarev, Inorganic Chemistry (Vysshaya shkola, Moscow, 1974), Vol. 2 [in Russian].
D. V. Korol’kov and G. A. Skorobogatov, The Fundamentals of Theoretical Chemistry (Akademiya, Moscow, 2004) [in Russian].
R. G. Pearson, Inorg. Chem. 27, 734 (1988).
C. K. Jørgensen, Prog. Inorg. Chem. 4(1), 73 (1962).
Author information
Authors and Affiliations
Additional information
Original Russian Text © D.E. Chirkst, O.L. Lobacheva, I.V. Berlinskii, N.V. Dzhevaga, 2012, published in Zhurnal Neorganicheskoi Khimii, 2012, Vol. 57, No. 4, pp. 670–674.
Rights and permissions
About this article
Cite this article
Chirkst, D.E., Lobacheva, O.L., Berlinskii, I.V. et al. Thermodynamics of formation of lanthanide hydroxo complexes in aqueous solutions. Russ. J. Inorg. Chem. 57, 605–609 (2012). https://doi.org/10.1134/S0036023612040067
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023612040067