Abstract
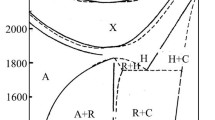
The phase compositions of theLaVO4-SrMoO4(1) and Sr2GeO4-SrMoO4 (2) binary systems, which bound the Sr2GeO4-LaVO4-SrMoO4 (3) ternary system, and the LaSr2(VO4)(GeO4)-Sr2GeO4+SrMoO4 section (4) of system 3 are studied at subsolidus temperatures. Systems 1 and 2 consist of a mixture of the initial compounds, and the La1 − x Sr2 + x (GeO4)(V1 − x Mo x O4) (where 0 ≤ x ≤ 0.4) region of substitutional solid solutions with a palmierite structure is formed in system 3. The unit cell parameters of the solid solutions are determined. The distribution of the lanthanum and strontium cations over two positions of the cationic sublattice is described.
Similar content being viewed by others
References
PDF2, Card 50-367.
G. Le Flem and R. Olasculaga, Bul. Soc. Chim. Fr., No. 7, 2769 (1968).
M. Escobar and Baran, Z. Anorg. Allg. Chem. 441, 273 (1978).
L. N. Dem’yanets, V. V. Ilyukhin, A. V. Chigarov, and N. V. Belov, Izv. Akad. Nauk SSSR, Neorg. Mater. 3(12), 2221 (1967).
E. Guermen, E. Daniels, and J. S. King, J. Chem. Phys. 55, 1093 (1971).
F. Nishi and Y. Takeuchi, Z. Kristallogr. 211, 607 (1996).
R. Heindl, G. Tary, and B. Blanzat, C.R. Seances Acad. Sci., Ser. 2, 310, 1037 (1990).
V. D. Zhuravlev, Yu. A. Velikodnyi, and M. Ya. Khodos, Zh. Neorg. Khim. 34(4), 1082 (1989).
A. C. Larson and R. V. Vjn Dreele, General Structure Analysis System (GSAS). Los Alamos Natl. Lab. Report LAUR, Nos. 86-748, 223 (2004).
V. S. Urusov, Energy Theory of Isomotrpous Miscibility (Nauka, Moscow, 1977) [in Russian].
J. M. S. Skakle, A. M. Coats, and J. Marr, J. Mater. Sci. 35, 3251 (2000).
Author information
Authors and Affiliations
Additional information
Original Russian Text © V.D. Zhuravlev, V.G. Zubkov, A.P. Tyutyunnik, Yu.A. Velikodnyi, N.D. Koryakin, 2009, published in Zhurnal Neorganicheskoi Khimii, 2009, Vol. 54, No. 1, pp. 135–137.
Rights and permissions
About this article
Cite this article
Zhuravlev, V.D., Zubkov, V.G., Tyutyunnik, A.P. et al. Crystal structures of La1 − x Sr2 + x (GeO4)(V1 − x Mo x O4) (x= 0−0.4) solid solutions. Russ. J. Inorg. Chem. 54, 134–136 (2009). https://doi.org/10.1134/S0036023609010227
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023609010227