Abstract
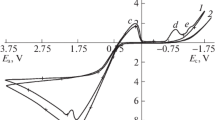
The dissolution of silver cathodically polarized in a 1M HC1 solution is investigated. It occurs only in the presence of oxygen, is independent of the cathodic potential in a range from 0.00 to −0.45 V, and is determined by the convective experimental conditions and the acidity of the solution. It may be assumed that silver can be oxidized at cathodic potentials because of an increase in the concentration of hydroxide ions adsorbed by the electrode, during the reduction of the dissolved oxygen.
Similar content being viewed by others
References
Florianovich, G.M., Zashch. Met., 2000, vol. 36, no. 10, p. 1175.
Kolotyrkin, Ya.M and Florianovich, G.M., Zashch. Met., 1984, vol. 20, no. 1, p. 14.
Zartsyn, I.D., Shugurov, A.E., and Marshakov, I.K., Zashch. Met., 2001, vol. 37, no. 2, p. 159.
Kreiser, I.V., Marshakov, I.K., Tutukina, N.M., and Zartsyn, I.D., Zashch. Met., 2004, vol. 40, no. 1, p. 28.
Altukhov, V.K. and Shatalov, V.G., Elektrokhimiya, 1987, vol. 23, no. 7, p. 968.
Spravochnik khimika (Chemical Handbook), Nikol’skii, B.P., Ed., Moscow-Leningrad, 1964, vol. 3, p. 140.
Kreizer, I.V., Tutukina, N.M., Zartsyn, I.D., and Marshakov, I.K., Zashch. Met., 2002, vol. 38, no. 3, p. 261.
Khimiya i tekhnologiya perekisi vodoroda (Chemistry and Technology of Hydrogen Peroxide), Seryshev, G.A., Ed., Leningrad, 1984.
Razumovskii, S.D., Kislorod-elementarnye formy i svoistva (Oxygen—Its Elementary Forms and Properties), Moscow: Khimiya, 1979.
Author information
Authors and Affiliations
Additional information
Original Russian Text © L.E. Volkova, I.K. Marshakov, N.M. Tutukina, I.V. Kreiser, 2006, published in Zashchita Metallov, 2006, Vol. 42, No. 2, pp. 154–157.
Rights and permissions
About this article
Cite this article
Volkova, L.E., Marshakov, I.K., Tutukina, N.M. et al. The dissolution of silver cathodically polarized in acid chloride media. Prot Met 42, 140–143 (2006). https://doi.org/10.1134/S0033173206020068
Received:
Issue Date:
DOI: https://doi.org/10.1134/S0033173206020068