Abstract
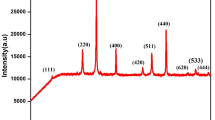
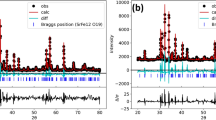
The composition and the structure of ceramic EuBa2Cu3O6 + δ (Eu-123) oxide samples annealed in steps with varying processing conditions (in air or oxygen and argon atmosphere at a temperature of 940–960°С for 1–70 h with or without homogenization) were studied by the X-ray phase and chemical analysis, electron diffraction pattern analysis, elemental analysis, and high-resolution transmission electron microscopy. Regardless of the processing conditions, Eu-123 nanostructured oxide with a tetragonal or orthorhombic structure and domains 1–20 nm in size was obtained as a result of annealing. Nanostructuring of the samples, which was revealed by high-resolution electron microscopy, is attributed to their chemical nature: the presence of identical structural elements in members of the homologous Eu n Ba m Cum + nO y series of oxides allows them to intergrow coherently and create an illusion of a single crystal. Just like any other member of the Eu n Ba m Cum + nO y series, oxide Eu-123 is disproportionate depending on the annealing conditions to form other members of this series located on either side of the dominant oxide. Temperature Tc of the superconducting transition of each member of the series depends on the average oxidation state of copper \(\overline {Cu} \). At \(\overline {Cu} \) < 2, all members of the series have a tetragonal structure and do not exhibit superconducting properties. At \(\overline {Cu} \) = 2.28, five members of the Eu n Ba m Cum + nO y series with matrices (Ba : Cu) 5 : 8, 3 : 5, 2 : 3, 5 : 7, and 3 : 4 exhibit superconducting properties with Tc = 82–90 K.
Similar content being viewed by others
References
L. A. Klinkova and V. I. Nikolaichik, “Nanostructural inhomogeneity of YBa2Cu3O7 - δ,” Physica C 506, 33–39.
L. A. Klinkova, V. I. Nikolaichik, N. V. Barkovskii, A. F. Shevchun, and V. K. Fedotov, “Existence of the homologous series of YnBamCum + nOy (m = 2, 3, 5; n = 1, 2) oxides with the tetragonal and orthorhombic structures of YBa2Cu3O6 + δ,” Russ. J. Inorg. Chem. 57, 1196–1209 (2012).
L. A. Klinkova, V. I. Nikolaichik, Q. M. Ramasse, and P. Abellan, “Local variations of cation composition on a nanometer-sized scale in a YBa2Cu3O6.92 superconductor,” J. Supercond. Novel Magn. 29, 1139–1143 (2016).
V. I. Nikolaichik and L. A. Klinkova, “Nanostructural Inhomogeneity of EuBa2Cu3O6 + δ Superconductors,” Bull. Russ. Acad. Sci.: Fiz. 80, 1418–1420 (2016).
L. A. Klinkova, V. I. Nikolaichik, N. V. Barkovskii, and V. K. Fedotov, “Primary crystallization field of the oxide EuBa2Cu3O6 + d and the existence of a homologous series EunBamCum + nOy (m = 2, 3, 4, 5; n = 1, 2),” Russ. J. Inorg. Chem. 60, 276–284 (2015).
T. Iwata, M. Hikita, Y. Tajima, and S. Tsurumi, “Study of the crystal structure of the high temperature superconductor Eu1Ba2Cu3O7 - y,” J. Cryst. Growth 85, 661–664 (1987).
S. A. Hodorowicz, W. Lasocha, A. Lasocha, and H. A. Eick, “The preparation and magnetic behavior of EuBa2Cu3 -yMyO6.5 + x (M = Ni, Co, Al, and Zn) and some related compounds,” J. Solid State Chem. 77, 148–155 (1988).
G. Bednorz, G. Stroink, and M.-A. White, “A Study of the heat capacity of the superconductor EuBa2Cu3O7–x,” Physica C 165, 385–390 (1990).
A. V. Dubovitskii, N. V. Kireev, N. D. Kushch, E. F. Makarov, M. K. Makova, A. T. Mailybaev, V. A. Merzhanov, S. I. Pesotskii, R. A. Stukan, and D. M. Shashkin, “Chemivam modification of the hightemperature superconductor Eu1Ba2Cu3O7 - δ with tin: Synthesis, structure, electrophysical properties, and Mössbauer studies,” Sverkhprovod.: Fiz., Khim., Tekh. 3, 1092–1103 (1990).
L. Malavasi, A. Tamburini, P. Galinetto, P. Ghigna, and Gl. Flor, “The high-temperature superconductor EuBa2Cu3O6 + x: Role of thermal history on microstructure and superconducting properties,” J. Mater. Synthes. Processing 9, 31–37 (2001).
E. Annese, G. Allodi, R. De Renzi, L. Malavasi, and P. Ghigna, “Phase Separation in Superconducting EuBa2Cu3O6 + x,” Physica B 326, 321–324.
Y. Li, Y. Liu, R. Duan, X. Xiong, B. Wang, G. Cao, L. Wei, D. N. Zheng, Z. X. Zhao, and J. H. Ross, “Positron annihilation study of the O-T phase transition for Eu1 + xBa2 -xCu3O7 - δ superconductors,” Physica C 402, 179–187.
Z. Chen, J. Zhang, Y. Su, Y. Xue, and G. Cao, “Effect of rare-earth ion size on local electron structure in RBa2Cu3O7 - δ (R = Tm, Dy, Gd, Eu, Nd and Y) superconductors: A positron study,” Physica C 434, 161–166.
E. I. Kuznetsova, Yu. V. Blinova, S. V. Sudareva, I. B. Bobylev, E. P. Romanov, and T. P. Krinitsina, “X-ray Diffraction Study of Spinodal Decomposition of a Nonstoichiometric Y–Ba–Cu–O Compound,” Phys. Met. Metallogr. 95, 65–70 (2003).
S. V. Sudareva, E. P. Romanov, T. P. Krinitsina, Yu. V. Blinova, and E. I. Kuznetsova, “Mechanism of Phase Transformations and Fine Structure of a Nonstoichiometric Compound YBa2Cu3O7–δ at Temperatures of 200 and 300°C,” Phys. Met. Metallogr. 102 (2), 205–212 (2006).
E. P. Romanov, S. V. Sudareva, E. N. Popova, and T. P. Krinitsina, Low-Temperatureand High-Tempera ture Suprconductors and Related Composites (UrO RAN, Ekaterinburg, 2009) [in Russian].
L. A. Klinkova, N. V. Barkovskii, S. S. Khasanov, and K. V. Van, “Study of the behavior of of the cuprates Ba2Cu3Oy and YBa2Cu3Oy in molten KOH,” Sverkhprovod.: Fiz., Khim., Tekh. 7, 377–384 (1994).
D. C. Harris and T. A. Hewston, “Determination of Cu3+/Cu2+ ratio in the superconductor YBa2Cu3O8–x,” J. Solid State Chem. 69, 182–185 (1987).
Y. Saito, T. Noji, K. Hirokawa, A. Endo, N. Matsuzaki, M. Katsumata, and N. Higuchi, “Determination of valence of copper and oxygen deficiency in superconducting SrxLa2–xCuO4–y,” Jpn. J. Appl. Phys. 26, L838–L839 (1987).
L. A. Klinkova, V. I. Nikolaichik, N. V. Barkovskii, and V. K. Fedotov, “On the existence of a homologous series of BamCum + nOy oxides with the cubic structure of the BaCuO2 oxide,” Physica C 470, 2067–2071 (2010).
ICSD No. 78467.
ICSD No. 65392.
M. Murakami, N. Sakai, T. Higuchi, and S. I. Yoo, “Melt-processed light rare earth element–Ba–Cu–O,” Supercond. Sci. Technol. 9, 1015–1032 (1996).
T. Egi, J. G. Wen, K. Kuroda, H. Unoki, and N. Koshizuka, “High critical-current density of Nd(Ba,Nd)2Cu3O7 - δ single crystals,” Appl. Phys. Lett. 67, 2406–2408 (1995).
W. Ting, T. Egi, R. Itti, K. Kuroda, N. Koshizuka, S. Tanaka, “Surface electronic properties and atomic images of the as-prepared Nd1Ba2Cu3Oy single crystals,” Jpn. J. Appl. Phys. 35, 4034–4037 (1996).
Yu. D. Tret’yakov and E. A. Gudilin, “Chemical principles of the preparation of metal-oxide superconductors,” Usp. Khim. 69, 3–40 (2000).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © L.A. Klinkova, V.I. Nikolaichik, N.V. Barkovskii, V.K. Fedotov, A.F. Shevchun, 2018, published in Fizika Metallov i Metallovedenie, 2018, Vol. 119, No. 3, pp. 232–242.
Rights and permissions
About this article
Cite this article
Klinkova, L.A., Nikolaichik, V.I., Barkovskii, N.V. et al. The Effect of Synthesis Conditions on the Phase Composition and Structure of EuBa2Cu3O6 + δ Samples. Phys. Metals Metallogr. 119, 216–228 (2018). https://doi.org/10.1134/S0031918X18030067
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0031918X18030067