Abstract
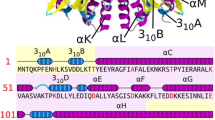
The structures of two crystal modifications of the W34F mutant ribonuclease from the bacterium Bacillus intermedius (binase) were solved and refined at 1.7 and 1.1 Å resolution. The kinetic parameters of the hydrolysis of substrates of different length (GpU, GpUp, and poly(I)) by binase and its W34F mutant were investigated and compared. The catalytic activity of the enzymes was shown to increase with increasing length of the substrate. The substitution of tryptophan for phenylalanine does not lead to a change in the activity of the enzyme but results in a decrease of the binding constants for substrates containing more than one phosphate groups. A comparison of the structure of the mutant enzyme with the previously established structures of binase and its complexes with sulfate ions and guanosine monophosphate showed that the difference in their kinetic parameters is related to the fact that the mutant ribonuclease cannot bind the second phosphate group. Both crystal modifications of the mutant binase contain dimers, like in the crystal structure of binase studied previously. In these dimers, only one enzyme molecule can bind the substrate molecule. Since the dimers were found in the crystals grown under four different conditions, it can be suggested that the enzyme can exist as dimers in solution as well. Mutants of binase, which could exclude the formation of dimers, are suggested.
Similar content being viewed by others
References
Hartley R.W. 1980. Homology between prokaryotic and eukaryotic ribonucleases. J. Mol. Evol. 15, 355–358.
Hill C., Dodson G., Heinemann U., Saenger W., Mitsui Y., Nakamura K., Borisov S., Tischenko G., Polyakov K., Pavlovsky A. 1983. The structural and sequence homology of microbial ribonucleases. Trends Biochem. Sci. 8, 364–369.
Heinemann U., Muller J.J., Polyakov K.M. 2000. Structure of ribonucleases. In: Landolt-Bornstein New series, 11/2a. Ed. Hinz H.J. Berlin: Springer, pp. 33–119.
Polyakov K.M., Lebedev A.A., Okorokov A.L., Panov K.I., Shulga A.A., Pavlovsky A.G., Karpeisky M.Ya., Dodson G.G. 2002. The structure of substrate-free microbial ribonuclease binase and its complex with 3’GMP and sulfate ions. Acta Crystallogr. Sect. D. 58, 744–750.
Day A.G., Parsonage D., Ebel S., Brown T., Ferst A.R. 1992. Barnase has subsites that give rise to large enhancements. Biochemistry. 31, 6390–6395.
Buckle A.M., Ferst A.R. 1994. Subsite binding in an RNase: Structure of a barnase-tetranucleotide complex at 1.76-Å resolution. Biochemistry. 33, 1644–1653.
Shul’ga A.A., Okorokov A.L., Panov K.I., Kurbanov F.T., Chernov B.K., Skryabin K.G., Kirpichnikov M.P. 1994. Overexpression in Bacillus intermedius 7P ribonuclease (binase) in Escherichia coli. Mol. Biol. 28, 303–310.
Kabsch W. 2001. Integration, scaling, space-group assignment and post refinement. XDS in International Tables for Crystallography. Eds. Rossmann M.G., Arnold E. Dordrecht: Kluwer, F, Chapter 25.2.9, pp. 730–734.
Vagin A., Teplyakov A. 1997. MOLREP: An automated program for molecular replacement. J. Appl. Cryst. 30, 1022–1025.
Murshudov G.N., Vagin A.A., Dodson E.J. 1997. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. Sect. D. 57, 240–255.
Emsley P., Cowtan K. Coot: Model-building tools for molecular graphics. 2004. Acta Crystallogr. Sect. D. 60, 2126–2132.
Ilinskaya O.N., Makarov A. A. 2005. Why ribonucleases induce tumor cell death. Mol. Biol. 39, 1–11.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © K.M. Polyakov, D.A. Goncharuk, A.A. Trofimov, T.N. Safonova, V.A. Mitkevich, E.N. Tkach, A.A. Makarov, A.A. Shulgad, 2010, published in Molekulyarnaya Biologiya, 2010, Vol. 44, No. 5, pp. 922–928.
Rights and permissions
About this article
Cite this article
Polyakov, K.M., Goncharuk, D.A., Trofimov, A.A. et al. X-ray diffraction and biochemical studies of W34F mutant ribonuclease binase. Mol Biol 44, 817–822 (2010). https://doi.org/10.1134/S0026893310050195
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026893310050195