Abstract
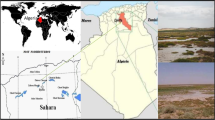
Admittedly, the Lut Desert of Iran has been remained as an unexplored region from a microbiological standpoint. Domain Archaea contains extremophiles that can live in harsh habitats. Extremely halophilic archaea are exposed to different environmental stresses in the hypersaline environments such as high solar irradiance and periodic desiccation. Haloarchaeal diversity in Shoor River, a saline river in the Lut Desert (a salinity of 134.3 g L–1 of dissolved salts), was investigated by a culture-dependent method. A large number of extremely halophilic isolates were obtained and a subset of 59 isolates was considered distinct. Firstly, the isolates were screened for their resistance under desiccation stress in 35 days. Eleven of these strains remained viable during the period in a desiccator containing silica gel. Then, three of them were randomly selected and their resistance against desiccation and ionizing radiation were determined. The isolates MS2, MS17, and MS50 were still recovered after 8 weeks in a desiccator and were moderately resistant to gamma radiation with D10 value between 2 and 3 kGy. Strains MS2, MS17, and MS50 were affiliated with three species in the family Halobacteriaceae using 16S rRNA gene sequence analysis as well as morphological and biochemical characteristics—Haloterrigena jeotgali A29T (99.6% similarity), Natrialba aegyptia 40T (99.4% similarity) and Natrinema pallidum NCIMB 777T (99.3% similarity), respectively. Although resistance to desiccation did not follow the sigmoid survival curve pattern of Deinococcus radiodurans, apparently haloarchaea can show a more resistance to desiccation in more long-term periods of time. This is the first report on isolation of extremely halophilic archaea belonged to the family Halobacteriaceae and their radioresistance and desiccation tolerance properties isolated from the Shoor River.
Similar content being viewed by others
References
Azua-Bustos, A., Urrejola, C., and Vicuna, R, Life at the dry edge: microorganisms of the Atacama Desert, FEBS Lett., 2012, vol. 586, no. 18, pp. 2939–2945.
Billi, D., Friedmann, E.I., Hofer, K.G., Caiola, M.G., and Ocampo-Friedmann, R., Ionizing-radiation resistance in the desiccation-tolerant cyanobacterium Chroococcidiopsis, Appl. Environ. Microbiol., 2000, vol. 66, no. 4, pp. 1489–1492.
Bull, A.T. and Asenjo, J.A, Microbiology of hyper-arid environments: recent insights from the Atacama Desert, Chile, Antonie van Leeuwenhoek, 2013, vol. 103, pp. 1173–1179.
Chanal, A., Chapon, V., Benzerara, K., Barakat, M., Christen, R., Achouak, W., Barras, F., and Heulin, T, The desert of Tataouine: an extreme environment that hosts a wide diversity of microorganisms and radiotolerant bacteria, Environ. Microbiol., 2006, vol. 8, pp. 514–525.
DeLong, E.F, Archaea in coastal marine environments, Proc. Natl. Acad. Sci. U. S. A., 1992, vol. 89, pp. 5685–5689.
Demergasso, C., Casamayor, E.O., Chong, G., Galleguillos, P., Escudero, L., and Pedros-Alio, C, Distribution of prokaryotic genetic diversity in athalassohaline lakes of the Atacama Desert, Northern Chile, FEMS Microbiol. Ecol., 2004, vol. 48, pp. 57–69.
Dussault, H.P, An improved technique for staining red halophilic bacteria, J. Bacteriol., 1995, vol. 70, pp. 484–485.
Dyall-Smith, M.L., The Halohandbook: Protocols for Haloarchaeal Genetics, 2006, pp. 10–15. http://www.haloarchaea.com/resources/halohandbook.
Felsenstein, J, Confidence limits on phylogenies: an approach using bootstrap, Evolution, 1985, vol. 39, pp. 783–791.
Fish, S.A., Shepherd, T.J., McGenity, T.J., and Grant, W.D, Recovery of 16S ribosomal RNA gene fragments from ancient halite, Nature, 2002, vol. 417, pp. 432–436.
Fredrickson, J.K., Li, S.M., Gaidamakova, E.K., Matrosova, V.Y., Zhai, M., Sulloway, H.M., Scholten, J.C., Brown, M.G., Balkwill, D.L., and Daly, M.J., Protein oxidation: key to bacterial desiccation resistance?, ISME J., 2008, vol. 2, pp. 393–403.
González, C., Gutiérrez, C., and Ramirez, C, Halobacterium vallismortis sp. nov., an amylolytic and carbohydratemetabolizing, extremely halophilic bacterium, Can. J. Microbiol., 1978, vol. 24, pp. 710–715.
Grant, W.D., Kamekura, M., McGenity, T.J., and Ventosa, A., Order I. Halobacteriales: the archaea and deeply branching and phototrophic bacteria, Bergey’s Manual of Systematic Bacteriology, Garrity, G.M., Ed., New York: Springer,2001, vol. 1.
Gtari, M., Essoussi, I., Maaoui, R., Sghaier, H., Boujmil, R., Gury, J., Pujic, P., Brusetti, L., Chouaia, B., Crotti, E., Daffonchio, D., Boudabous, A., and Normand, P, Contrasted resistance of stone-dwelling Geodermatophilaceae species to stresses known to give rise to reactive oxygen species, FEMS Microbiol. Ecol., 2012, vol. 80, pp. 566–577.
Hezayen, F.F., Rehm, B.H.A., Tindall, B.J., and Steinbüchel, A, Transfer of Natrialba asiatica B1T to Natrialba taiwanensis sp. nov. and description of Natrialba aegyptiaca sp. nov., a novel extremely halophilic, aerobic, non-pigmented member of the Archaea from Egypt that produces extracellular poly(glutamic acid), Int. J. Syst. Evol. Microbiol., 2001, vol. 51, pp. 1133–1142.
Jolivet, E., L’Haridon, S., Corre, E., Forterre, P., and Prieur, D, Thermococcus gammatolerans sp. nov., a hyperthermophilic archaeon from a deep-sea hydrothermal vent that resists ionizing radiation, Int. J. Syst. Evol. Microbiol., 2003, vol. 53, pp. 847–851.
Kish, A., Kirkali, G., Robinson, C., Rosenblatt, R., Jaruga, P., Dizdaroglu, M., and DiRuggiero, J, Salt shield: intracellular salts provide cellular protection against ionizing radiation in the halophilic archaeon, Halobacterium salinarum NRC-1, Environ. Microbiol., 2009, vol. 11, no. 5, pp. 1066–1078.
Kottemann, M., Kish, A., Iloanusi, C., Bjork, S., and DiRuggiero, J, Physiological responses of the halophilic archaeon Halobacterium sp. strain NRC1 to desiccation and gamma irradiation, Extremophiles, 2005, vol. 9, pp. 219–227.
Lane, D.J., Pace, B., Olsen, G.J., Stahl, D.A., Sogin, M.L., and Pace, N.R, Rapid determination of 16S ribosomal RNA sequences for phylogenetic analyses, Proc. Natl. Acad. Sci. U. S. A., 1985, vol. 82, pp. 6955–6959.
Makhalanyane, T.P., Valverde, A., Gunnigle, E., Frossard, A., Ramond, J.B., and Cowan, D.A, Microbial ecology of hot desert edaphic systems, FEMS. Microbiol. Rev., 2015, vol. 39, pp. 203–221.
Mattimore, V. and Battista, J.R, Radioresistance of Deinococcus radiodurans: functions necessary to survive ionizing radiation are also necessary to survive prolonged desiccation, J. Bacteriol., 1996, vol. 178, pp. 633–637.
McGenity, T.J., Gemmell, R.T., and Grant, W.D, Proposal of a new halobacterial genus Natrinema gen. nov., with two species Natrinema pellirubrum norn. nov. and Natrinerna pallidum norn. nov, Int. J. Syst. Evol. Microbiol., 1998, vol. 48, pp. 1187–1196.
McGenity, T.J., Gemmell, R.T., Grant, W.D., and Stan-Lotter, H, Origins of halophilic microorganisms in ancient salt deposits, Environ. Microbiol., 2000, vol. 2, no. 3, pp. 243–250.
Mohseni, M., Abbaszadeh, J, and Nasrollahi Omran, A., Radiation resistant of native Deinococcus spp. isolated from the Lout desert of Iran “the hottest place on Earth,” Int. J. Environ. Sci. Technol., 2014, vol. 11, pp. 1939–1946.
Oren, A., Life at High Salt Concentrations, in The Prokaryotes. A Handbook on the Biology of Bacteria, Dworkin, M., Ed., Springer,2006, vol. 2, pp. 263–282.
Oren, A, Microbial life at high salt concentrations: phylogenetic and metabolic diversity, Saline Systems., 2008, vol. 4, no. 2, pp. 1–13.
Oren, A., Ventosa, A., and Grant, W.D, Proposed minimal standards for description of new taxa in the order Halobacteriales, Int. J. Syst. Bacteriol., 1997, vol. 47, pp. 233–238.
Rainey, F.A., Ray, K., Ferreira, M., Gatz, B.Z., Nobre, M.F., Bagaley, D., Rash, B.A., Park, M.J., Earl, A.M., Shank, N.C., Small, A.M., Henk, M.C., Battista, J.R., Kämpfer, P., and da Costa, M.S, Extensive diversity of ionizing-radiation-resistant bacteria recovered from Sonoran Desert soil and description of nine new species of the genus Deinococcus obtained from a single soil sample, Appl. Environ. Microbiol., 2005, vol. 71, no. 9, pp. 5225–5235.
Robinson, C.K., Webb, K., Kaur, A., Jaruga, P., Dizdaroglu, M., Baliga, N.S., Place, A., and DiRuggiero, J., A major role for nonenzymatic antioxidant processes in the radioresistance of Halobacterium salinarum, J. Bacteriol., 2011, vol. 193, no. 7, pp. 1653–1662.
Roh, S.W., Nam, Y.D., Chang, H.W., Kim, K.H., Sung, Y., Kim, M.S., Oh, H.M., and Bae, J.W, Haloterrigena jeotgali sp. nov., an extremely halophilic archaeon from salt-fermented food, Int. J. Syst. Evol. Microbiol., 2009, vol. 59, no. 9, pp. 2359–2363.
Saitou, N. and Nei, M, The neighbor-joining method: a new method for reconstructing phylogenetic trees, Mol. Biol. Evol., 1987, vol. 4, pp. 406–425.
Shahmohammadi, H.R., Asgarani, E., Terato, H., Saito, T., Ohyama, Y., Gekko, K., Yamamoto, O., and Ide, H, Protective roles of bacterioruberin and intracellular KCl in the resistance of Halobacterium salinarium against DNA-damaging agents, J. Radiat. Res., 1998, vol. 39, no. 4, pp. 251–262.
Slade, D. and Radman, M, Oxidative stress resistance in Deinococcus radiodurans, Microbiol. Mol. Biol. Rev., 2011, vol. 75, pp. 133–191.
Smibert, R.M. and Krieg, N.R., Phenotypic characterization, in Methods for General and Molecular Bacteriology, Gerhardt, P., Murray, R.G.E., Wood, W.A., and Krieg, N.R., Eds., Washington: Amer. Soc. Microbiol., 1994, pp. 607–654.
Tamura, K., Stecher, G., Peterson, D., Filipski, A., and Kumar, S., MEGA6: Molecular Evolutionary Genetics Analysis version 6.0., Mol. Biol. Evol., 2013, vol. 30, pp. 2725–2729.
Tanaka, M., Earl, A.M., Howell, H.A., Park, M.J., Eisen, J.A., Peterson, S.N., and Battista, J.R, Analysis of Deinococcus radiodurans’s transcriptional response to ionizing radiation and desiccation reveals novel proteins that contribute to extreme radioresistance, Genetics, 2004, vol. 168, no. 1, pp. 21–33.
Thompson, J.D., Gibson, T.J., Plewniak, F., Jeanmougin, F., and Higgins, D.J, The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools, Nucleic. Acids. Res., 1997, vol. 25, pp. 4876–4882.
Ventosa, A., Unusual microorganisms from unusual habitats: hypersaline environments, in Prokaryotic Diversity–Mechanisms and Significance, Logan, N.A., Lappin-Scott, H.M., and Oyston, P.C.F., Eds., Cambridge Univ. Press., 2006, pp. 223–254.
Webb, K.M. and DiRuggiero, J, Role of Mn2+ and compatible solutes in the radiation resistance of thermophilic bacteria and archaea, Archaea, 2012, pp. 1–11.
Yazdi, A., Emami, M.H., and Shafiee, S.M., Dasht-e Lut in Iran, the most complete collection of beautiful geomorphological phenomena of desert, Open J. Geol., 2014, vol. 4, pp. 249–261.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Shirsalimian, M.S., Amoozegar, M.A., Sepahy, A.A. et al. Isolation of extremely halophilic Archaea from a saline river in the Lut Desert of Iran, moderately resistant to desiccation and gamma radiation. Microbiology 86, 403–411 (2017). https://doi.org/10.1134/S0026261717030158
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026261717030158