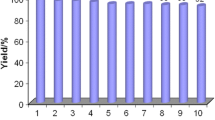
Abstract
Bis(indolyl)alkanes are a class of alkaloids that possess significant biological activities. Every year, the varieties of bis(indolyl)alkanes isolated from natural sources are increasing. Nevertheless, the deficiency of natural products from natural sources led to a decrease in the exploration of natural products for biological investigations. Corresponding to this fact, there is a demand to develop efficient protocols for the construction of bis(indolyl)alkanes. In this regards, we have prepared phospho sulfonic acid (PSA), which is a non-corrosive, highly reactive, inexpensive and low toxic catalyst, and have applied to construct bis(indolyl)alkanes. The catalyst was prepared by reaction of diammonium hydrogen phosphate with chloro sulfuric acid and was fully characterized by FTIR spectrometry. PSA has been used as a solid acid catalyst to promote the electrophilic substitution reaction of indole with aldehydes under water to furnish a library of bis(indolyl)alkanes in excellent yields over short reaction times. The present method eliminates use of toxic catalyst and solvent, and tolerates a series of functional groups. The catalyst can be reused several times without any important activity loss. The methodology has several advantages, for example, simple experimental procedure, cost efficiency (use of inexpensive catalyst), ease of preparation and handling of the catalyst and no side reactions.




Similar content being viewed by others
REFERENCES
Praveen, I.J., Parameswaran, P.S., and Majik, M.S., Synthesis, 2015, vol. 47, no. 13, p. 1827.
Shiri, M., Zolfigol, M.A., Kruger, H.G., and Tanbakouchian, Z., Chem. Rev., 2009, vol. 110, no. 4, p. 2250.
Safe, S., Papineni, S., and Chintharlapalli, S., Cancer Lett., 2008, vol. 269, p. 326.
Anderton, M.J., Manson, M.M., Verschoyle, R., Gescher, A., Steward, W.P., Williams, M.L., and Mager, D.E., Drug Metab. Dispos., 2004, vol. 32, p. 632.
Pisano, C., Kollar, P., Gianni, M., Kalac, Y., Giordano, V., Ferrara, F.F., Tancredi, R., Devoto, A., Rinaldi, A., Rambaldi, A., Penco, S., Marzi, M., Moretti, G., Vesci, L., Tinti, O., Carminati, P., Terao, M., and Garattini, E., Blood, 2002, vol. 100, p. 3719.
Parkin, D.R., Lu, Y.J., Bliss, R.L., and Malejka-Giganti, D., Food Chem. Toxicol., 2008, vol. 46, p. 2451.
Bell, R., Carmeli, S., and Sar, N., J. Nat. Prod., 1994, vol. 57, p. 1587.
Kamal, A., Khan, M.N.A., Reddy, K.S., Srikanth, Y.V.V., Ahmed, S.K., Kumar, K.P., and Murthy, U.S.N., J. Enzyme Inhib. Med. Chem., 2009, vol. 24, p. 559.
Mahboobi, S., Teller, S., Pongratz, H., Hufsky, H., Sellmer, A., Botzki, A., Uecker, A., Beckers, T., Baasner, S., Schaechtele, C., Ueberall, F., Kassack, M.U., Dove, S., and Boehmer, F.-D., J. Med. Chem., 2002, vol. 45, p. 1002.
Gong, Y., Firestone, G.L., and Bjeldanes, L.F., Mol. Pharmacol., 2006, vol. 69, p. 1320.
Chen, I., McDougal, A., Wang, F., and Safe, S., Carcinogenesis, 1998, vol. 19, p. 1631.
Garikapaty, V.P.S., Ashok, B.T., Tadi, K., Mittelman, A., and Tiwari, R.K., Biochem. Biophys. Res. Commun., 2006, vol. 340, p. 718.
Sun, S.S., Han, J., Ralph, W.M., Chandrasekaran, A., Liu, K., Auborn, K.J., and Carter, T.H., Cell Stress Chaperones, 2004, vol. 9, p. 76.
Nachshon-Kedmi, M., Yannai, S., and Fares, F.A., Br. J. Cancer, 2004, vol. 91, p. 1358.
McDougal, A., Gupta, M.S., Morrow, D., Ramamoorthy, K., Lee, J.E., and Safe, S.H., Breast Cancer Res. Treat., 2001, vol. 66, p. 147.
Sung, D.C., Yoon, K., Chintharlapalli, S., Abdelrahim, M., Lei, P., Hamilton, S., Khan, S., Ramaiah, S.K., and Safe, S., Cancer Res., 2007, vol. 67, p. 674.
Ichite, N., Chougule, M.B., Jackson, T., Fulzele, S.V., Safe, S., and Singh, M., Clin. Cancer Res., 2009, vol. 15, p. 543.
Inamoto, T., Papineni, S., Chintharlapalli, S., Cho, S.D., Safe, S., and Kamat, A.M., Mol. Cancer Ther., 2008, vol. 7, p. 3825.
Sujatha, K., Perumal, P.T., Muralidharan, D., and Rajendran, M., Indian J. Chem., 2009, vol. 48B, p. 267.
Kirkus, M., Tsai, M.H., Grazulevicius, J.V., Wu, C.-C., Chi, L.C., and Wong, K.T., Synth. Met., 2009, vol. 159, p. 729.
Nagarajan, R. and Perumal, P.T., Chem. Lett., 2004, vol. 33, no. 3, p. 288.
Faisal, M., Hussain, S., Haider, A., Saeed, A., and Larik, F.A., Chem. Pap., 2018, vol. 3, p. 1.
Hasaninejad, A., Zare, A., Sharghi, H., Niknam, K., and Shekouhy, M., ARKIVOC, 2007, vol. 14, p. 39.
Hasaninejad, A., Zare, A., Sharghi, H., Shekouhy, M., Khalifeh, R., Salimi Beni, A., and Moosavi Zare, A.R., Can. J. Chem., 2007, vol. 85, p. 416.
Hasaninejad, A., Parhami, A., Zare, A., Khalafi Nezhad, A., Nasrolahi Shirazi, A., and Moosavi Zare, A.R., Pol. J. Chem., 2008, vol. 82, p. 565.
Koshima, H. and Matsuaka, W., J. Heterocycl. Chem., 2002, vol. 39, p. 1089.
Bandgar, B.P. and Shaikh, K.A., Tetrahedron Lett., 2003, vol. 44, p. 1959.
Zare, A., Parhami, A., Moosavi-Zare, A.R., Hasaninejad, A., Khalafi-Nezhad, A., and Beyzavi, M.H., Can. J. Chem., 2009, vol. 87, p. 416.
Shiri, M., Zolfigol, M.A., Kruger, H.G., and Tanbakouchian, Z., Chem. Rev., 2010, vol. 110, p. 2250.
Yadav, J.S., Reddy, B.V.S., Murthy, C.V.S.R., Kumar, G.M., and Madan, C., Synthesis, 2001, vol. 5, p. 783.
Firouzabadi, H., Iranpour, N., Jafarpour, M., and Ghaderi, A., J. Mol. Cat. A: Chem., 2006, vol. 253, p. 249.
Yadav, J.S., Reddy, B.V.S., Satheesh, G., Prabhakar, A., and Kunwar, A.C., Tetrahedron Lett., 2003, vol. 44, p. 2221.
Boroujeni, K.P. and Parvanak, K., Chin. Chem. Lett., 2011, vol. 22, p. 939.
Yang, Q., Yin, Z.L., Ouyang, B.L., and Peng, Y.Y., Chin. Chem. Lett., 2011, vol. 22, p. 515.
Jafarpour, M., Rezaeifard, A., and Golshani, T., J. Heterocycl. Chem., 2009, vol. 46, p. 535.
Sadaphal, S.A., Kategaonkar, A.H., Labade, V.B., and Shingare, M.S., Chin. Chem. Lett., 2010, vol. 21, p. 39.
Ji, S.J., Zhou, M.F., Gu, D.G., Jiang, Z.Q., and Loh, T.P., Eur. J. Org. Chem., 2004, vol. 2004, p. 1584.
Hosseini-Sarvari, M., Synth. Commun., 2008, vol. 38, p. 832.
Yadav, J.S., Reddy, B.V.S., Padmavani, B., and Gupta, M.K., Tetrahedron Lett., 2004, vol. 45, p. 7577.
Kokare, N.D., Sangshetti, J.N., and Shinde, D.B., Chin. Chem. Lett., 2008, vol. 19, p. 1186.
Alinezhad, H., Haghighi, A.H., and Salehian, F., Chin. Chem. Lett., 2010, vol. 21, p. 183.
Azizian, J., Teimouri, F., and Mohamadizadeh, M.R., Catal. Commun., 2007, vol. 8, p. 1117.
Zhang, C.L. and Du, Z.Q., Chin. Chem. Lett., 2009, vol. 20, p. 1411.
Chakrabarty, M., Ghosh, N., Basaka, R., and Harigaya, Y., Tetrahedron Lett., 2002, vol. 43, p. 4075.
Kobayashi, S., Araki, M., and Yasuda, M., Tetrahedron Lett., 1995, vol. 6, p. 5773.
Faisal, M., Shahid, S., Ghumro, S.A., Saeed, A., Larik, F.A., Shaheen, Z., Channar, P.A., Fattah, T.A., Rasheed, S., and Mahesar, P.A., Synth. Commun., 2018, vol. 48, no. 4, p. 462.
Faisal, M., Saeed, A., Shahzad, D., Fattah, T.A., Lal, B., Channar, P.A., Mahar, J., Saeed, S., Mahesar, P.A., and Larik, F.A., Eur. J. Med. Chem., 2017, vol. 141, p. 386.
Faisal, M., Saeed, A., Larik, F.A., Ghumro, S.A., Rasheed, S., and Channar, P.A., J. Electron. Mater., 2018, vol. 47, no. 12, p. 7011.
Naidu, K.R.M., Khalivulla, S.I., Kumar, P.C.R., and Lasekan, O., Org. Commun., 2012, vol. 5, no. 3, p. 150.
Shaikh, A.C., and Chen, C., J. Chin. Chem. Soc. (Taipei), 2011, vol. 58, no. 7, p. 899.
Zare, A., Parhami, A., Moosavi-Zare, A.R., Hasaninejad, A., Khalafi-Nezhad, A., and Beyzavi, M.H., Can. J. Chem., 2008, vol. 87, no. 2, p. 416.
Kalla, R.M.N., Hong, S.C., and Kim, I., ACS Omega, 2018, vol. 3, no. 2, p. 2242.
Yang, Y., Xie, Z., and Wang, J., Chin. J. Chem., 2011, vol. 29, no. 10, p. 2091.
Vahdat, S.M., Khaksar, S., and Baghery, S., World Appl. Sci. J., 2012, vol. 19, p. 1003.
Peng, Y.Y., Zhang, Q.L., Yuan, J.J., and Cheng, J.P., Chin. J. Chem., 2008, vol. 26, no. 12, p. 2228.
Hosseini-Sarvari, M., Acta Chim. Slov., 2007, vol. 54, no. 2, p. 354.
Simha, P.R., Mangali, M.S., Kuppireddy Gari, D., Venkatapuram, P., and Adivireddy, P., J. Heterocycl. Chem., 2017, vol. 54, no. 5, p. 2717.
Naidu, K.R.M., Khalivulla, S.I., Kumar, P.C.R., and Lasekan, O., Org. Commun., 2012, vol. 5, no. 3, p. 150.
Li, J.T., Zhang, X.H., and Song, Y.L., Int. J. Chem. Tech. Res., 2010, vol. 2, no. 1, p. 341.49.
Kiasat, R.A., Mouradezadegun, A., and Saghanezhad, J.S., J. Serb. Chem. Soc., 2013, vol. 78, no. 4, p. 469.
Kalla, R.M.N., Park, H., Hoang, T.T.K., and Kim, I., Tetrahedron Lett., 2014, vol. 55, no. 39, p. 5373.
Rezayati, S., Erfani, Z., and Hajinasiri, R., Chem. Pap., 2015, vol. 69, no. 4, p. 536.
Kiasat, A.R. and Hemat-Alian, L., Res. Chem. Intermed., 2015, vol. 41, no. 2, p. 873.
Rezayati, S., Mehmannavaz, M., Salehi, E., Haghi, S., Hajinasiri, R., and Afshari Sharif Abad, S., J. Sci. I. R. Iran, 2016, vol. 27, no. 1, p. 51.
Kiasat, R.A., Mouradezadegun, A., and Saghanezhad, J.S., J. Serb. Chem. Soc., 2013, vol. 78, no. 4, p. 469.
Kalla, R.M.N., Lee, H.R., Cao, J., Yoo, J.W., and Kim, I., New J. Chem., 2015, vol. 39, no. 5, p. 3916.
Faisal, M., Larik, F.A., and Saeed, A., J. Porous Mater., 2018, vol. 26, no. 2, p. 455.
Simon, M.O. and Li, C.J., Chem. Soc. Rev., 2012, vol. 41, no. 4, p. 1415.
Chanda, A. and Fokin, V.V., Chem. Rev., 2009, vol. 109, no. 2, p. 725.
Hasaninejad, A., Shekouhy, M., Zare, A., Ghattali, S.H., and Golzar, N., J. Iran. Chem. Soc., 2011, vol. 8, no. 2, p. 411.
Staskun, B., J. Org. Chem., 1964. vol. 29, p. 1153.
Shaabani, A., Soleimani, E., and Badri, Z., Synth. Commun., 2007, vol. 37, p. 629.
Yadav, J.S., Reddy, B.V.S., Sreedhar, P., Rao, R.S., and Nagaiah, K., Synthesis, 2004, p. 2381.
Firouzabadi, H., Iranpoor, N., and Jafari, A.A., J. Mol. Catal. A: Chem., 2006, vol. 244, p. 168.
Wang, L., Han, J.H., Sheng, T., Fan, J.Z., and Tang, X., Synlett, 2005, p. 337.
Magesh, C.J., Nagarajan, R., Karthik, M., and Perumal, P.T., Appl. Catal., A, 2004, vol. 266, p. 1.
Nagarajan, R., and Perumal, P.T., Synth. Commun., 2002, vol. 32, p. 105.
Niknam, K., Zolfigol, M.A., Sadabadi, T., and Nejati, A., J. Iran. Chem. Soc., 2006, vol. 3, p. 318.
Nagarajan, R. and Perumal, P.T., Tetrahedron, 2000, vol. 58, p. 1229.
Chen, D.P., Yu, L.B., and Wang, P.G., Tetrahedron Lett., 1996, vol. 37, p. 4467.
Firouzabadi, H., Iranpour, N., Jafarpour, M., and Ghaderi, A., J. Mol. Catal. A: Chem., 2006, vol. 253, p. 249.
Zare, A., Parhami, A., Moosavi-Zare, A.R., Hasaninejad, A., Khalafi-Nezhad, A., and Beyzavi, M.H., Can. J. Chem., 2009, vol. 87, p. 416
Khalafi-Nezhad, A., Parhami, A., Zare, A., Moosavi Zare, A.R., Hasaninejad, A., and Panahi, F., Synthesis, 2008, p. 617.
Hasaninejad, A., Zare, A., Sharghi, H., Niknam, K., and Shekouhy, M., ARKIVOC, 2007, vol. 14, p. 39.
Hasaninejad, A., Parhami, A., Zare, A., Khalafi Nezhad, A., Nasrolahi Shirazi, A., and Moosavi Zare, A.R., Pol. J. Chem., 2008, vol. 82, p. 565.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Faisal, M., Larik, F.A., Salman, M. et al. Phospho Sulfonic Acid: A Highly Efficient and Novel Catalyst for Formation of Bis(Indolyl)Alkanes from Aldehydes and Indole under Aqueous Conditions. Kinet Catal 60, 522–535 (2019). https://doi.org/10.1134/S0023158419040049
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158419040049