Abstract
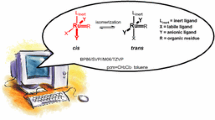
The hydroisomerization of a cis-isomer to produce a trans-isomer on Rh, Pd, Pt, Ru, and Ir/α-Al2O3 catalysts is studied. It is shown that Rh and Ru catalysts on which the hydroisomerization reaction mostly takes place exhibit the most favorable characteristics, whereas on the other metals, the main route is the hydrogenation reaction. Rh/α-Al2O3 is the optimum catalyst, since it has much higher activity than Ru/α-Al2O3. It is found that the increased selectivity of the trans-isomer formation is facilitated by a decrease in the hydrogen pressure and by an increase in the substrate concentration. The maximum selectivity is achieved when the reaction is carried out in nonpolar n-hexane and toluene, whereas in the case of the more polar tetrahydrofuran (THF), dimethylformamide (DMFA), and methanol both the reaction rate and the selectivity of the trans-isomer formation decline.
Similar content being viewed by others
References
Paust, J. and John, M., US Patent 5689022, 1997.
Nicolaou, K.C., Daines, R.A., Chakraborty, T.K., and Ogawa, Y., J. Am. Chem. Soc., 1987, vol. 109, p. 2821.
Semenov, V.V., Kiselyov, A.S., Titov, I.Y., Sagamanova, I.K., Ikizalp, N.N., Chernysheva, N.B., Tsyganov, D.V., Konyushkin, L.D., Firgang, S.I., Semenov, R.V., Karmanova, I.B., Raihstat, M.M., and Semenova, M.N., J. Nat. Prod., 2010, vol. 73, p. 1796.
Mori, K., In Total Synthesis of Natural Products, Ed. J. ApSimon, New York: Wiley Interscience, 1992, vol. 9, p. 521.
Mori, K., Top. Curr. Chem., 2004, vol. 239, p. 1.
Odinokov, V.N., Chem. Nat. Compd., 2000, vol. 36, p. 11.
Chen, M. and Roush, W.R., Org. Lett., 2012, vol. 14, p. 1880.
Fouché M., Rooney, L. and Barrett, A.G.M., Org. Chem., 2012, vol. 77, p. 3060.
Oger, C., Balas, L., Durand T., and Galano J.-M, Chem. Rev., 2013, vol. 113, p. 1313.
Ananikov, V.P., Khokhlova, E.A., Egorov, M.P., Sakharov, A.M., Zlotin, S.G., Kucherov, A.V., Kustov, L.M., Gening, M.L., and Nifantiev, N.E., Mendeleev Commun., 2015, vol. 25, p. 75.
Keitz, B.K., Endo, K., Patel, P.R., Herbert, M.B., and Grubbs, R.H., J. Am. Chem. Soc., 2012, vol. 134, p. 693.
Grubbs, R.H., Handbook of Metathesis, Wiley-CH: Weinheim, Germany, 2003, p. 1234.
Trost, B.M., Ball, Z.T., and Joge, T., J. Am. Chem. Soc., 2002, vol. 124, p. 7922.
Wittig, G. and Schollkopf, U., Chem. Ber., 1954, vol. 87, p. 1318.
Maryanoff, B.E. and Reitz, A.B., Chem. Rev., 1989, vol. 89, p. 863.
Wadsworth, W. and Emmons, W.D., J. Am. Chem. Soc., 1961, vol. 83, p. 1733.
Blakemore, P.R., J. Chem. Soc., Perkin Trans., 2002, vol. 1, p. 2563.
Peterson, D.J., Org. Chem., 1968, vol. 33, p. 780.
Ananikov, V.P., Galkin, K.I., Egorov, M.P., Sakharov, A.M., Zlotin, S.G., Redina, E.A., Isaeva, V.I., Kustov, L.M., Gening, M.L., and Nifantiev, N.E., Mendeleev Commun., 2016, vol. 26, p. 365.
Karunananda, M.K. and Mankad, N.P., J. Am. Chem. Soc., 2015, vol. 137, p. 14598.
Neumann, K.T., Klimczyk, S., Burhardt, M.N., Bang-Andersen, B., Skrydstrup, T., and Lindhardt, A.T., ACS Catal., 2016, vol. 6, p. 4710.
Kusy, R. and Grela, K., Org. Lett., 2016, vol. 18, p. 6196.
Komatsu, T., Takagi, K., and Ozawa, K., Catal. Today, 2011, vol. 164, p. 143.
Furukawa, S., Yokoyama, A., and Komatsu, T., ACS Catal., 2014, vol. 4, p. 3581.
Kachala, V.V., Khemchyan, L.L., Kashin, A.S., Orlov, N.V., Grachev, A.A., Zalesskii, S.S., and Ananikov, V.P., Russ. Chem. Rev., 2013, vol. 82, p. 648.
Markov, P.V., Bragina, G.O., Baeva, G.N., Tkachenko, O.P., Mashkovskii, I.S., Yakushev, I.A., Kozitsyna, N.Yu., Vargaftik, M.N, and Stakheev, A.Yu., Kinet. Catal., 2015, vol. 56, p. 591.
Bond, G.C., Metal-Catalyzed Reactions of Hydrocarbons, Springer: Rickmansworth, UK, 2005, p. 666.
Kogan, V.B., Fridman, V.M., and Kafarov, V.V., Spravochnik po rastvorimosti. Binarnye sistemy (Handbook of Solubility. Binary Systems), Kafarov, V.V., Eds., vol. 1, no. 1, Moscow: Izd-vo ANSSSR, 1961, p. 959.
Furukawa, S., Ochi, K., Luo, H., Miyazaki, M., and Komatsu, T., Chem. Cat. Chem., 2015, vol. 7, p. 3472.
Studer, M., Baumeister, P., Blaser, H., and Bach, J., J. Mol. Catal. A, 1995, vol. 95, p. 155.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © P.V. Markov, I.S. Mashkovsky, G.N. Baeva, A.Yu. Stakheev, 2017, published in Kinetika i Kataliz, 2017, Vol. 58, No. 6, pp. 751–759.
Rights and permissions
About this article
Cite this article
Markov, P.V., Mashkovsky, I.S., Baeva, G.N. et al. Hydroisomerization of cis-Stilbene into trans-Stilbene on Supported Heterogeneous Metal Catalysts (Rh, Pd, Pt, Ru, Ir/α-Al2O3). Kinet Catal 58, 771–779 (2017). https://doi.org/10.1134/S0023158417060064
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158417060064