Abstract
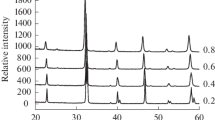
Radically different dependences of the activity of La1 − x Sr x MnO3 (x = 0−0.5) perovskites in methane oxidation on the degree of substitution of strontium for lanthanum are observed for low and high temperatures. Unsubstituted LaMnO3 exhibits the highest activity in the temperature range from 300 to 500°C, while the sample with the maximum degree of substitution (La0.5Sr0.5MnO3) shows the highest activity at higher temperatures of 700–900°C. In the low temperature region, the activity of La1t - x Sr x MnO3 is determined by the amount of weakly bound (overstoichiometric) oxygen, which is formed in cation-deficient lattices and is characterized by a thermal desorption peak with T max = 705°C. At higher temperatures (800–900°C), the strongly bound oxygen of the catalyst lattice is involved in the formation of the reaction products under both unsteady- and steady-state conditions. As a consequence, the catalytic activity in methane oxidation correlates with the apparent rate constant of oxygen diffusion in the oxide bulk.
Similar content being viewed by others
References
Boreskov, G.K., Sazonov, V.A., and Popovskii, V.V., Dokl. Akad. Nauk SSSR, 1967, vol. 176, p. 1331.
Popovskii, V.V., Boreskov, G.K., Muzykantov, V.S., Sazonov, V.A., and Shubnikov, S.G., Kinet. Katal., 1969, vol. 10, p. 786.
Popovskii, V.V., Kinet. Katal., 1972, vol. 13, p. 1190.
Yakovleva, I.S., Isupova, L.A., Rogov, V.A., and Sadykov, V.A., Kinet. Katal., 2008, vol. 49, no. 2, p. 274 [Kinet. Catal. (Engl. Transl.), vol. 49, no. 2, p. 261].
Isupova, L.A., Yakovleva, I.S., Alikina, G.M., Rogov, V.A., and Sadykov, V.A., Kinet. Katal., 2005, vol. 46, p. 773 [Kinet. Catal. (Engl. Transl.), vol. 46, p. 729].
Marchetti, L. and Forni, L., Appl. Catal., B, 1998, vol. 15, p. 179.
Alifanti, M., Kirchnerova, J., Delmon, B., and Klvana, D., Appl. Catal., A, 2004, vol. 262, p. 167.
Nitadori, T., Kurihara, S., and Misono, M., J. Catal., 1986, vol. 98, p. 221.
Choudhary, T.V., Banerjee, S., and Choudhary, V.R., Appl. Catal., A, 2002, vol. 234, p. 1.
Tejuca, L.G., Fierro, J.L.G., and Tascon, J.M.D., Adv. Catal., 1989, vol. 36, p. 237.
Yamazoe, N. and Teraoka, Y., Catal. Today, 1990, vol. 8, p. 175.
Sadykov, V.A., Kuznetsova, T.G., Simakov, A.V., Rogov, V.A., Zaikovskii, V.I., Moroz, E.M., Kochubei, D.I., Novgorodov, B.N., Ivanov, V.P., Trukhan, S.N., Litvak, G.S., Bulgakov, N.N., Lunin, V.V., and Kemnitz, E., Mater. Res. Soc. Symp. Proc., 2003, vol. 751.
Happel, J., Isotopic Assessment of Heterogeneous Catalysis, Orlando, Fla.: Academic, 1986.
US Patent 3330697, 1967.
Ivanov, D.V., Pinaeva, L.G., Isupova, L.A., Nadeev, A.N., Prosvirin, I.P., and Dovlitova, L.S., Catal. Lett. (in press).
Shannon, S.L. and Goodwin, J.G., Jr., Chem. Rev., 1995, vol. 95, p. 677.
Ivanov, D.V., Sadovskaya, E.M., Pinaeva, L.G., and Isupova, L.A., J. Catal., 2009, vol. 257, p. 5.
Bal’zhinimaev, B.S. and Pinaeva, L.G., Kinet. Katal., 1995, vol. 36, p. 60.
Rentgenografiya (X-Ray Crystallography), Katsnel’son, A.A., Ed., Moscow: Mosk. Gos. Univ., 1986, p. 237.
Beznosikov, B.V. and Aleksandrov, K.S., Kristallografiya, 2000, vol. 45, no. 5, p. 864 [Crystallogr. Rep. (Engl. Transl.), vol. 45, no. 5, p. 792].
Mitchel, J.F., Argyriou, D.N., Potter, C.D., Hinks, D.G., Jorgensen, J.D., and Bader, S.D., Phys. Rev. B: Condens. Matter, 1996, vol. 54, p. 6172.
Seiyama, T., Yamazoe, N., and Eguchi, K., Ind. Eng. Chem. Res., 1995, vol. 24, p. 19.
Ponce, S., Pena, M.A., and Fierro, J.L.G., Appl. Catal., B, 2000, vol. 24, p. 193.
Muzykantov, V.S., Popovskii, V.V., and Boreskov, G.K., Kinet. Katal., 1964, vol. 5, p. 624.
Taskin, A. and Lavrov, A.N., Yoichi Ando, Appl. Phys. Lett., 2005, vol. 86, p. 0919110.
Opila, E.J., Tuller, H.I., Wuensch, B.J., and Maier, J., J. Am. Ceram. Soc., 1993, vol. 76, p. 2363.
Borovskikh, L., Mazo, G., and Kemnitz, E., Solid State Sci., 2003, vol. 5, p. 409.
Lee, Y.N., Lago, R.M., Fierro, J.L., Cortes, V., Sapina, F., and Martinez, E., Appl. Catal., A, 2001, vol. 207, p. 17.
Jinguang Deng, Yue Zhang, Hongxing Dai, Lei Zhang, Hong He, and Au, C.T., Catal. Today, 2008, vol. 139, p. 82.
Lavasseur, B. and Kaliaguine, S., Appl. Catal., A, 2008, vol. 343, p. 29.
Sadykov, V.A., Bulgakov, N.N., Muzykantov, V.S., Kuznetsova, T.G., Alikina, G.M., Lukashevich, A.I., Potapova, Yu.V., Rogov, V.A., Burgina, E.B., Zaikovskii, V.I., Moroz, E.M., Litvak, G.S., Yakovleva, I.S., Isupova, L.A., Zyryanov, V.V., Kemnitz, E., and Neophytides, S., in Mixed Ionic Electronic Conducting Perovskites for Advanced Energy Systems, Orlovskaya, N. and Browning, N., Eds., NATO Science Series II: Mathematics, Physics and Chemistry, 2003, vol. 173, p. 53.
Vazhnova, T.G., Korchak, V.N., Krylov, O.V., Slin’ko, M.M., Kinet. Katal., 1985, vol. 26, p. 1378.
Petrolekas, P.D. and Matcalfe, I.S., J. Catal., 1995, vol. 152, p. 143.
Ramzi Hammani, Salwa Ben Aissa, and Habib Batis, Appl. Catal., A, 2009, vol. 353, p. 145.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © D.V. Ivanov, L.G. Pinaeva, E.M. Sadovskaya, L.A. Isupova, 2011, published in Kinetika i Kataliz, 2011, Vol. 52, No. 3, pp. 410–418.
Rights and permissions
About this article
Cite this article
Ivanov, D.V., Pinaeva, L.G., Sadovskaya, E.M. et al. Influence of the mobility of oxygen on the reactivity of La1 − x Sr x MnO3 perovskites in methane oxidation. Kinet Catal 52, 401–408 (2011). https://doi.org/10.1134/S0023158411030086
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158411030086