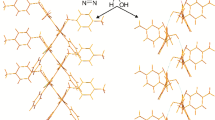
Abstract
We confirm our previously formulated rule about the possibility of a spontaneous resolution of enantiomers of a series of 5-hydroxy-3-pyrrolin-2-one thioderivatives. Crystal packings of three racemic compounds are compared. A supramolecular associate of the same type (a hydrogen-bonded dimer) is found, which is differently packed in the 3D structure of the crystals.












Similar content being viewed by others
REFERENCES
P. A. Levkin, V. Y. Torbeev, D. A. Lenev, and R. G. Kostyanovski. Top. Stereochem., 2006, 25, 81. https://doi.org/10.1002/0471785156.ch4
J. Jacques, A. Collet, and S. H. Wilen. Enantiomers, Racemates and Resolutions. Krieger Publishing Company, 1994.
L. Perez-Garcia and D. B. Amabilino. Chem. Soc. Rev., 2007, 36, 941. https://doi.org/10.1039/B610714A
D. P. Gerasimova, A. F. Saifina, D. V. Zakharychev, A. R. Zaripova, R. R. Fayzullin, A. R. Kurbangalieva, and O. A. Lodochnikova. J. Struct. Chem., 2021, 62(5), 727. https://doi.org/10.1134/S0022476621050097
O. A. Lodochnikova, A. R. Zaripova, R. R. Fayzullin, A. I. Samigullina, I. I. Vandyukova, L. N. Potapova, and A. R. Kurbangalieva. CrystEngComm, 2018, 20, 3218. https://doi.org/10.1039/C8CE00369F
D. P. Gerasimova, A. F. Saifina, D. V. Zakharychev, R. R. Fayzullin, A. R. Kurbangalieva, and O. A. Lodochnikova. CrystEngComm, 2021, 23, 3907. https://doi.org/10.1039/D1CE00227A
D. P. Gerasimova, O. A. Lodochnikova, A. R. Kurbangalieva, and I. A. Litvinov. J. Struct. Chem., 2021, 62(9), 1425. https://doi.org/10.1134/S0022476621090110
O. A. Lodochnikova, Y. K. Voronina, L. Z. Latypova, D. B. Krivolapov, A. R. Kurbangalieva, and I. A. Litvinov. Russ. Chem. Bull., 2013, 62, 1218. https://doi.org/10.1007/s11172-013-0167-1
D. P. Gerasimova, R. R. Fayzullin, D. V. Zakharychev, A. F. Saifina, I. I. Vandyukova, A. R. Kurbangalieva, and O. A. Lodochnikova. Abstracts of Reports: X National Crystal Chemical Conference. Elbrus region, Russia, July 5-9, 2021. Chernogolovka, Russia: IPCP RAS, 2021, 80. [In Russian]
O. A. Lodochnikova, L. S. Kosolapova, A. F. Saifina, A. T. Gubaidullin, R. R. Fayzullin, A. R. Khamatgalimov, I. A. Litvinov, and A. R. Kurbangalieva. CrystEngComm, 2017, 19, 7277. https://doi.org/10.1039/C7CE01717K
D. P. Gerasimova, A. F. Saifina, D. V. Zakharychev, I. I. Vandyukova, R. R. Fayzullin, A. R. Kurbangalieva, and O. A. Lodochnikova. J. Struct. Chem., 2020, 61(3), 476. https://doi.org/10.1134/S0022476620030142
L. Krause, R. Herbst-Irmer, G. M. Sheldrick, and D. Stalke. J. Appl. Crystallogr., 2015, 48, 3. https://doi.org/10.1107/S1600576714022985
G. M. Sheldrick. Acta Crystallogr., Sect. A: Found. Adv., 2015, 71, 3. https://doi.org/10.1107/S2053273314026370
G. M. Sheldrick. Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71, 3. https://doi.org/10.1107/S2053229614024218
O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard, and H. J. Puschmann. J. Appl. Crystallogr., 2009, 42, 339. https://doi.org/10.1107/S0021889808042726
L. J. Farrugia. J. Appl. Crystallogr., 2012, 45, 849. https://doi.org/10.1107/S0021889812029111
A. J. Gordon and R. A. Ford. The Chemists Companion: a Handbook of Practical Data, Techniques, and References. Wiley, 1972.
H. Simonis. Chem. Ber., 1901, 34, 509. https://doi.org/10.1002/cber.19010340184
L. Cao, S.-H. Luo, H.-Q. Wu, L.-Q. Chen, K. Jiang, Z.-F. Hao, and Z.-Y. Wang. Adv. Synth. Catal., 2017, 359, 2961. https://doi.org/10.1002/adsc.201700600
Funding
Physicochemical studies were carried out in the Multi-Access Spectral Analytical Center of the Kazan Scientific Center, Russian Academy of Sciences and supported by the State Assignment of the Kazan Scientific Center, Russian Academy of Sciences.
The synthesis of the key compounds was supported by the Kazan Federal University Strategic Academic Leadership Program (Priority-2030).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interests.
Additional information
Russian Text © The Author(s), 2022, published in Zhurnal Strukturnoi Khimii, 2022, Vol. 63, No. 10, 99529.https://doi.org/10.26902/JSC_id99529
Rights and permissions
About this article
Cite this article
Gerasimova, D.P., Faizova, R.G., Zakharychev, D.V. et al. STABILITY AND REPRODUCIBILITY OF THE DIMERIC MOTIF IN THE CRYSTALS OF THIOETHERS OF 3-BROMO-5-HYDROXY-1- (4-METHYLBENZYL)-1,5-DIHYDRO-2H-PYRROL-2-ONES. J Struct Chem 63, 1616–1628 (2022). https://doi.org/10.1134/S0022476622100080
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476622100080