Abstract
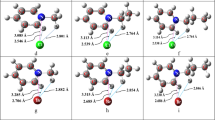
Structural and thermodynamic characteristics of molecular and ionic complexes of aluminum trichloride with pyrazine (pyz) and 4,4′-bipyridyl (bipy) are calculated at the RI-BP86/def2-SVP level. It is found that for molecular 2AlCl3·3L and 4AlCl3·3L complexes an energy difference between isomers does not exceed 4 kJ/mol, and the rotation barrier of the AlCl3 moiety relative to the N-Al-N bond does not exceed 24 kJ/mol. A comparison of the stability of molecular and ionic complexes of aluminum in the gas phase shows that the maximum energy difference is ∼60 kJ/mol. For L = pyz the molecular complex is more stable whereas for L = bipy it is the ionic one.
Similar content being viewed by others
References
A. Y. Timoshkin and H. F. Schaefer, Chem. Rec., 2, No. 5, 319 (2002).
E. I. Davydova, T. N. Sevastianova, A. V. Suvorov, et al., Coord. Chem. Rev., 254, No. 17–18, 2031 (2010).
C. R. Samanamu, P. M. Lococo, and A. F. Richards, Inorg. Chim. Acta, 360, No. 14, 4037 (2007).
J. L. Atwood, F. R. Bennett, C. Jones, et al., J. Chem. Soc., Chem. Comm., No. 7, 541 (1992).
G. J. Palenik, Acta Crystallogr., 17, No. 12, 1573 (1964).
S. P. Petrosyants and A. B. Ilyukhin, Neorg. Khim., 55, No. 1, 30 (2010).
R. Restivo and G. J. Palenik, J. Chem. Soc., Dalton Trans., No. 3, 341 (1972).
A. Sofetis, C. P. Raptopoulou, A. Terzis, et al., Inorg. Chim. Acta, 359, No. 10, 3389 (2006).
R. Ahlrichs, M. Bär, M. Häser, et al., Chem. Phys. Lett., 162, No. 3, 165 (1989).
A. D Becke., Phys. Rev. A, 38, No. 6, 3098 (1988).
J. P. Perdew, Phys. Rev. B, 33, No. 12, 8822 (1986).
K. Eichkorn, O. Treutler, H. Öhm, et al., Chem. Phys. Lett., 242, No. 6, 652 (1995).
K. Eichkorn, F. Weigend, O. Treutler, and R. Ahlrichs, Theor. Chem. Acc., 97, Nos. 1–4, 119 (1997).
F. Weigend, Phys. Chem. Chem. Phys., 8, No. 9, 1057 (2006).
F. Weigend and R. Ahlrichs, Phys. Chem. Chem. Phys., 7, No. 18, 3297 (2005).
A. Schäfer, H. Horn, and R. Ahlrichs, J. Chem. Phys., 97, No. 4, 2571 (1992).
E. Z. Zasorin and N. G. Rambidi, J. Struct. Chem., 8, No. 3, 347–352 (1967).
P. Pullmann, K. Hensen, and J. W. Bats, Z. Naturforsch. B, 37, No. 10, 1312 (1982).
N. C. Means, C. M. Means, S. G. Bott, et al., Inorg. Chem., 26, No. 9, 1466 (1987).
N. R. Strel’nikova, V. K. Bel’skii, L. V. Ivakina, et al., Koordinats. Khim., 12, No. 8, 1101 (1987).
P. Shukla, J. C. Gordon, A. H. Cowley, et al., J. Organomet. Chem., 690, No. 5, 1366 (2005).
S. A. Cortes-Llamas, Z. Garcia-Hernandez, E. Rufino-Felipe, et al., Inorg. Chim. Acta, 363, No. 9, 3959 (2010).
N. C. Baenziger, Acta Crystallogr., 4, No. 3, 216 (1951).
NIST Chemistry WebBook, http://webbook.nist.gov/chemistry/
T. N. Sevast’yanova and A. V. Suvorov, Koordinats. Khim., 25, No. 10, 679 (1999).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © 2014 E. I. Davydova, A. Yu. Timoshkin, G. Frenking.
__________
Translated from Zhurnal Strukturnoi Khimii, Vol. 55, No. 1, pp. 20–28, January–February, 2014.
Rights and permissions
About this article
Cite this article
Davydova, E.I., Timoshkin, A.Y. & Frenking, G. Structure and stability of molecular and ionic complexes of AlCl3 with pyrazine and 4,4′-bipyridyl. J Struct Chem 55, 15–22 (2014). https://doi.org/10.1134/S002247661401003X
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002247661401003X