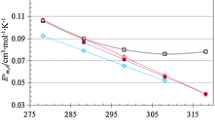
Abstract
The dependence of the standard partial volumes of glycine, α-alanine, and serine on the ionic strength of aqueous sodium chloride and sulfate solutions is modeled by the extended Masson equation: {ie534-1}. The error of less than 0.2 cm3/mol is a result of using five values of the A and B parameters: the two values of A are determined by the type of salt and the three values of B by the type of amino acid. A new variation of the additive-group approach is proposed for {ie534-2}. The partial volumes of the CH3 group (α-alanine) and the CH2 group (serine) are found not to depend on the salt concentration. The partial volume of the CH2 group of glycine grows with concentration. The structural characteristics of the hydrated complexes of the NH +3 and COO− groups are calculated: the hydration numbers, the molar volumes of water inside and outside the hydration sphere, and the intrinsic volume of NH +3 in COO− in solution. Given the same ionic strength, the aqueous sodium sulfate solution produces a somewhat stronger dehydration of the charged groups.
Similar content being viewed by others
References
Q. Yuan, Z.-F. Li, and B.-H. Wang, J. Chem. Thermodyn., 38, 20–33 (2006).
S. K. Singh and N. Kishore, J. Sol. Chem., 32, 117–135 (2003).
Y. Marcus, J. Sol. Chem., 23, 831–848 (1994).
J. V. Leyendekkers, J. Chem. Faraday Trans. I, 79, 1123–1134 (1983).
M. N. Islam and R. K. Wadi, J. Bangladesh Chem. Soc., 7, 206–211 (1994).
R. K. Wadi and P. Ramasami, J. Chem. Faraday Trans., 93, 243–247 (1997).
M. Ide, Y. Maeda, and H. Kitano, J. Phys. Chem. B, 101, 7022–7026 (1997).
V. P. Korolev, J. Struct. Chem., 51, 491–499 (2010).
R. S. Humphrey, G. R. Hedwig, I. D. Watson, and G. N. Malcolm, J. Chem. Thermodyn., 12, 595–603 (1980).
Z. Yan, J. Wang, W. Liu, and J. Lu, Thermochim. Acta, 334, 17–27 (1999).
T. Ogawa, K. Mizutani, and M. Yasuda, Bull.Chem. Soc. Jpn., 57, 2064–2068 (1984).
C. Liu and C. Ren, J. Chem. Eng. Data, 54, 3296–3299 (2009).
Gy. Jákli, J. Chem. Thermodyn., 39, 1589–1600 (2007).
C. M. Romero and J. C. Cadena, J. Sol. Chem., 39, 1474–1483 (2010).
A. V. Hakin and J. L. Liu, J. Sol. Chem., 35, 1157–1171 (2006).
J. E. Desnoyers, M. Arel, G. Perron, and C. Jolicoeur, J. Phys. Chem., 73, 3346–3351 (1969).
G. Perron, N. Desrosiers, and J. E. Desnoyers, Canad. J. Chem., 54, 2163–2183 (1976).
A. K. Mishra, K. P. Prasad, and J. C. Ahluwalia, Biopolymers, 22, 2397–2409 (1983).
A. K. Mishra and J. C. Ahluwalia, J. Phys. Chem., 88, 86–92 (1984).
V. P. Korolev, J. Struct. Chem., 52, 737–742 (2011).
V. P. Korolev and A. L. Serebryakova, J. Struct. Chem., 52, 1106–1110 (2011).
L. Lepori and P. Gianni, J. Sol. Chem., 29, 405–447 (2000).
A. Lo Surdo, E. M. Alzola, and F. J. Millero, J. Chem. Thermodyn., 14, 649–662 (1982).
J. C. Hindman, J. Chem. Phys., 36, 1000–1015 (1962).
J. V. Leyendekkers, J. Phys. Chem., 90, 5449–5455 (1986).
A. Zavitsas, J. Phys. Chem. B, 105, 7805–7817 (2001).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text Copyright © 2013 by V. P. Korolev
__________
Translated from Zhurnal Strukturnoi Khimii, Vol. 54, No. 3, pp. 482–487, May–June, 2013
Rights and permissions
About this article
Cite this article
Korolev, V.P. Volumetric properties, structural effects, and hydration of amino acids in aqueous salt solutions. J Struct Chem 54, 534–540 (2013). https://doi.org/10.1134/S0022476613030098
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476613030098