Abstract
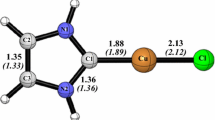
A series of N-heterocyclic carbene nickel complexes of the type [Ni(N-heterocylic carbene)(NO)(R)] (R = H, Me, HC=CH2, and C≡CH) are examined to study the influence of a substituent on the molecular structure and bonding of these complexes. Geometrical and AIM analyses of the interaction between Ni and the carbene fragment reveal that for the metal-carbene bond donation is more important than back-donation. The NICS values suggest that aromaticity in the heterocyclic ring is less than in the free heterocycle.
Similar content being viewed by others
References
F. E. Hahn and M. C. Jahnke, Angew. Chem., Int. Ed., 47, 3122 (2008).
W. A. Herrmann, Angew. Chem., Int. Ed., 41, 1290 (2002).
M. Scholl, S. Ding, C. W. Lee, and R. H. Grubbs, Org. Lett., 1, 953 (1999).
C. Boehme and G. Frenking, Organometallics, 17, 5801 (1998).
M.-T. Lee and C.-H. Hu, Organometallics, 23, 976 (2004).
J. Schwarz, V. P. W. Bohm, M. G. Gardiner, M. G. Gardiner, M. Grosche, W. A. Herrmann, W. Hieringer, and G. Raudaschl-Sieber, Chem. Eur. J., 6, 1773 (2000).
T. Weskamp, F. J. Kohl, W. Hieringer, D. Gleich, and W. A. Herrmann, Angew. Chem., Int. Ed., 38, 2416 (1999).
M. Tafipolsky, W. Scherer, K. O. Fele, G. Artus, B. Pedersen, W. A. Herrmann, and G. S. McGrady, J. Am. Chem. Soc., 124, 5865 (2002).
M. S. Varonka and T. H. Warren, Organometallics, 29, 717 (2010).
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, J. A. Montgomery Jr., T. Vreven, K. N. Kudin, J. C. Burant, J. M. Millam, S. S. Iyengar, J. Tomasi, V. Barone, B. Mennucci, M. Cossi, G. Scalmani, N. Rega, G. A. Petersson, H. Nakatsuji, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, M. Klene, X. Li, J. E. Knox, H. P. Hratchian, J. B. Cross, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, P. Y. Ayala, K. Morokuma, G. A. Voth, P. Salvador, J. J. Dannenberg, V. G. Zakrzewski, S. Dapprich, A. D. Daniels, M. C. Strain, O. Farkas, D. K. Malick, A. D. Rabuck, K. Raghavachari, J. B. Foresman, J. V. Ortiz, Q. Cui, A. G. Baboul, S. Clifford, J. Cioslowski, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. L. Martin, D. J. Fox, T. Keith, M. A. Al-Laham, C. Y. Peng, A. Nanayakkara, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, C. Gonzalez, and J. A. Pople, Revision B.03 Ed.; Gaussian, Inc., Pittsburgh PA (2003).
R. Ditchfield, W. J. Hehre, and J. A. Pople, J. Chem. Phys., 54, 724 (1971).
P. Hariharan and J. A. Pople, Mol. Phys., 27, 209 (1974).
M. S. Gordon, Chem. Phys. Lett., 76, 163 (1980).
P. J. Hay and W. R. Wadt, J. Chem. Phys., 82, 299 (1985).
P. J. Hay and W. R. Wadt, J. Chem. Phys., 82, 284 (1985).
A. Schaefer, H. Horn, and R. Ahlrichs, J. Chem. Phys., 97, 2571 (1992).
P. J. Hay and W. R. Wadt, J. Chem. Phys., 82, 270 (1985).
A. D. Becke, J. Chem. Phys., 104, 1040 (1996).
C. Adamo and V. Barone, Chem. Phys. Lett., 274, 242 (1997).
A. E. Reed, L. A. Curtiss, and F. Weinhold, Chem. Rev., 88, 899 (1988).
W. Zierkiewicz, D. Michalska, and P. Hobza, Chem. Phys. Lett., 386, 95 (2004).
D. Michalska, W. Zierkiewicz, D. C. Bien’ko, W. Wojciechowski, and T. Zeegers-Huyskens, J. Phys. Chem. A, 105, 8734 (2001).
P. M. Wojciechowski, W. Zierkiewicz, D. Michalska, and P. Hobza, J. Chem. Phys., 118, 10900 (2003).
P. v. R. Schleyer, C. Maerker, A. Dransfeld, H. Jiao, and N. J. R. v. E. Hommes, J. Am. Chem. Soc., 118, 6317 (1996).
Z. Chen, C. S. Wannere, C. Corminboeuf, R. Puchta, and P. v. R. Schleyer, Chem. Rev., 105, 3842 (2005).
R. F. W. Bader, Ver 2.0, Ed. Hamilton, McMaster University (2000).
R. G. Parr, L v.S.l.,and S. Liu, J. Am. Chem. Soc., 121, 1922 (1999).
R. G. Pearson, Chemical Hardness, Wiley, VCH, Oxford (1997).
R. G. Parr and W. Yang, Density-Functional Theory of Atoms and Molecules, Oxford University Press, New York (1989).
G. Frenking, M. Sola, and S. F. Vyboishchikov, J. Organometallic Chem., 690, 6178 (2005).
M. Cases, G. Frenking, M. Duran, and M. Sola, Organometallics, 21, 4182 (2002).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text Copyright © 2012 by R. Ghiasi
The text was submitted by the authors in English. Zhurnal Strukturnoi Khimii, Vol. 53, No. 2, pp. 376–380, March–April, 2012.
Rights and permissions
About this article
Cite this article
Ghiasi, R. Chemical bonding and properties in [Ni(N-heterocylic carbene)(NO)(R)] (R = H, Me, HC=CH2, and C≡CH) complexes: Theoretical insights. J Struct Chem 53, 377–382 (2012). https://doi.org/10.1134/S0022476612020254
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476612020254