Abstract
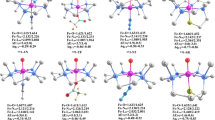
A DFT/B3LYP method using the 6-311++G(3df) basis set is employed to calculate the geometric, electronic, and thermodynamic parameters of O=NO-ON=O peroxide as an isomer of N2O4 dinitrogen tetraoxide. Calculations of the configuration interaction in a system of three paramagnetic particles with open shells have shown for the first time that the formation of cis-cis peroxide in the oxidation reaction of nitrogen oxide 2NO (2Π) + O2 (3Σg) → O=NO-ON=O (1 A) proceeds without an energy barrier in accordance with recently performed studies. The molecular orbital scheme of the barrierless activation of molecular oxygen and the driving force of the NO oxidation reaction are considered. A spontaneous character of the process is based on the idea of spin-catalysis when the reaction proceeds in the two-triplet state with total zero spin. The obtained results are in agreement with the experimental data on a spontaneous and irreversible process characterized by the observed negative activation energy.
Similar content being viewed by others
References
H. Tsukahara, T. Ishida, and M. Mayumi, Nitric Oxide, 3, No. 3, 191–198 (1999).
M. L. McKee, J. Am. Chem. Soc., 117, 1629–1637 (1995).
L. P. Olson, K. T. Kuwata, M. D. Bartberger, et. al., J. Am. Chem. Soc., 124, No. 32, 9469–9475 (2002).
O. B. Gadzhiev, S. K. Ignatov, A. G. Razuvaev et. al., J. Phys. Chem., A113, No. 32, 9092–9101 (2009).
V. I. Atroshchenko, A. M. Alekseev, A. P. Zasorin, et al., Technology of Bound Nitrogen [in Russian], Vishcha Shkola, Kiev (1985).
T. G. Akhmetov (ed.), in: Chemical Technology of Inorganic Compounds [in Russian], Vysshaya Shkola, Moscow (2002).
W. A. Guillory and H. S. Johnston, J. Chem. Phys., 42, 2457–2561 (1965).
S. C. Bhatia and J. H. Hall, J. Phys. Chem., 84, No. 24, 3255–3259 (1980).
I. I. Zakharov, V. F. Anufrienko, O. I. Zakharova, et al., J. Struct. Chem., 46, No. 2, 212 (2005).
P. E. M. Siegbahn, J. Comput. Chem., 6, 182–188 (1985).
K. Mikhaylichenko, C. Riehn, L. Valachovic, et al., J. Chem. Phys., 105, 6807–6817 (1996).
E. D. Morris and H. S. Johnston, J. Chem. Phys., 47, 4282 (1967).
W. Eisfeld and K. Morokuma, J. Chem. Phys., 119, 4682–4688 (2003).
I. I. Zakharov and O. I. Zakharova, J. Struct. Chem., 50, No. 2, 212–218 (2009).
X. Wang and Q.-Z. Qin, Int. J. Quant. Chem., 76, 77–82 (2000).
I. I. Zakharov, A. I. Kolbasin, O. I. Zakharova, et al., Theor. Experim. Chem., 44, No. 1, 26–31 (2008).
G. R. Smith and W. A. Guillory, J. Mol. Spectroscop., 68, 223–235 (1977).
A. D. Becke, Phys. Rev., A38, 3098–3100 (1988).
C. Lee, W. Yang, and R. G. Parr, Phys. Rev., B37, 785–789 (1988).
M. J. Frisch, G. W. Trucks, H. B. Schlegel, et al., Gaussian 03, Revision B.02, Gaussian, Inc., Pittsburgh PA (2003).
L. Noodleman, J. Chem. Phys., 74, 5737–5743 (1981).
J. J. P. Stewart, J. Comp. Chem., 10, No. 2, 209–220 (1989).
B. F. Minaev, Usp. Khim., 76, No. 11, 1039–1063 (2007).
I. I. Zakharov, B. F. Minaev, D. V. Petrenko, et al., Visn. Cherkas’kogo Universitetu, 174, 49–59 (2010).
B. F. Minaev, J. Mol. Catalysis A, 171, Nos. 1/2, 53–72 (2001).
Author information
Authors and Affiliations
Additional information
Original Russian Text Copyright © 2012 by I. I. Zakharov and B. F. Minaev
__________
Translated from Zhurnal Strukturnoi Khimii, Vol. 53, No. 1, pp. 7–17, January–February, 2012.
Rights and permissions
About this article
Cite this article
Zakharov, I.I., Minaev, B.F. A quantum chemical study of the structure of O=NO-ON=O peroxide and the reaction mechanism of no oxidation in the gas phase. J Struct Chem 53, 1–11 (2012). https://doi.org/10.1134/S0022476612010015
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476612010015