Abstract
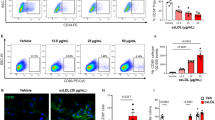
Endothelial dysfunction is among the major triggers of cardiovascular disease, also promoting thrombosis, restenosis, and neoatherosclerosis within the conduit and coronary artery upon the coronary artery bypass graft surgery. To recapitulate the paracrine interactions between arterial and venous endothelial cells (ECs) by cross-addition of the conditioned medium to the respective cell cultures. Conditioned (24 hours) serum-free medium from confluent primary human coronary artery ECs (HCAEC), primary human internal thoracic artery ECs (HITAEC) and primary human saphenous vein ECs (HSaVEC) was added to a confluent monolayer of intact HCAEC, HITAEC, and HSaVEC for another 24 hours. Cellular response has been assessed by reverse transcription-quantitative polymerase chain reaction, Western blotting and enzyme-linked immunosorbent assay. Gene expression analysis showed that HITAEC-conditioned medium ameliorated inflammatory response in HCAEC, in contrast to HSaVEC-conditioned medium. In turn, HCAEC-conditioned medium maintained the endothelial phenotype of HITAEC. Further, HCAEC- and HITAEC-conditioned medium stimulated release of pro-angiogenic molecules by intact HCAEC and HSaVEC cultures. HSaVEC-conditioned medium notably reduced the secretion of pro-inflammatory cytokine interleukin-6 in all cell cultures. Conclusions: Paracrine interactions between HITAEC and HCAEC govern their anti-inflammatory status and enhance HCAEC angiogenic potential, partially explaining the higher primary patency of arterial conduits and resistance of coronary artery to thrombosis, restenosis and neoatherosclerosis in the long-term period after total arterial revascularisation.





Similar content being viewed by others
REFERENCES
dela Paz NG, D’Amore PA (2009) Arterial versus venous endothelial cells. Cell Tissue Res 335(1): 5–16. https://doi.org/10.1007/s00441-008-0706-5
Rafii S, Butler JM, Ding BS (2016) Angiocrine functions of organ-specific endothelial cells. Nature 529(7586): 316–325. https://doi.org/10.1038/nature17040
Verrier ED, Boyle EM Jr (1996) Endothelial cell injury in cardiovascular surgery. The Ann Thorac Surg 62(3): 915–922. https://doi.org/10.1016/s0003-4975(96)00528-0
Augustin HG, Koh GY (2017) Organotypic vasculature: From descriptive heterogeneity to functional pathophysiology. Science (New York) 357(6353): eaal2379. https://doi.org/10.1126/science.aal2379
Zeff RH, Kongtahworn C, Iannone LA, Gordon DF, Brown TM, Phillips SJ, Skinner JR, Spector M (1988) Internal mammary artery versus saphenous vein graft to the left anterior descending coronary artery: prospective randomized study with 10-year follow-up. Ann Thorac Surg 45(5): 533–536. https://doi.org/10.1016/s0003-4975(10)64526-2
Ray FR, Huang W, Slater M, Barden JA (2002) Purinergic receptor distribution in endothelial cells in blood vessels: a basis for selection of coronary artery grafts. Atherosclerosis 162(1): 55–61. https://doi.org/10.1016/S0021-9150(01)00681-5
Wadey K, Lopes J, Bendeck M, George S (2018) Role of smooth muscle cells in coronary artery bypass grafting failure. Cardiovasc Res 114(4): 601–610. https://doi.org/10.1093/cvr/cvy021
Hadinata IE, Hayward PA, Hare DL, Matalanis GS, Seevanayagam S, Rosalion A, Buxton BF (2009) Choice of conduit for the right coronary system: 8-year analysis of Radial Artery Patency and Clinical Outcomes trial. Ann Thorac Surg 88(5): 1404–1409. https://doi.org/10.1016/j.athoracsur.2009.06.010
Lytle BW, Loop FD, Cosgrove DM, Ratliff NB, Easley K, Taylor PC (1985) Long-term (5 to 12 years) serial studies of internal mammary artery and saphenous vein coronary bypass grafts. J Thorac Cardiovasc Surg 89(2): 248–258.
Shishkova D, Markova V, Sinitsky M, Tsepokina A, Frolov A, Zagorodnikov N, Bogdanov L, Kutikhin A (2020) Co-Culture of Primary Human Coronary Artery and Internal Thoracic Artery Endothelial Cells Results in Mutually Beneficial Paracrine Interactions. Int J Mol Sci 21(21): 8032. https://doi.org/10.3390/ijms21218032
Dimeling G, Bakaeen L, Khatri J, Bakaeen FG (2021) CABG: When, why, and how? Cleve Clin J Med 88(5): 295–303. https://doi.org/10.3949/ccjm.88a.20115
Alizadehghobadi S, Biglari H, Niroomand-Oscuii H, Matin MH (2021) Numerical study of hemodynamics in a complete coronary bypass with venous and arterial grafts and different degrees of stenosis. Comput Methods Biomech Biomed Engin 24(8): 883–896. https://doi.org/10.1080/10255842.2020.1857744
Frolov AV (2019) Morphological and functional system of graft-artery junctions. Complex Issues of Cardiovascular Diseases 8(1): 112–122. https://doi.org/10.17802/2306-1278-2019-8-1-112-122
Gaudino M, Antoniades C, Benedetto U, Deb S, Di Franco A, Di Giammarco G, Fremes S, Glineur D, Grau J, He GW, Marinelli D, Ohmes LB, Patrono C, Puskas J, Tranbaugh R, Girardi LN, Taggart DP, ATLANTIC (Arterial Grafting International Consortium) Alliance (2017) Mechanisms, Consequences, and Prevention of Coronary Graft Failure. Circulation 136(18): 1749–1764. https://doi.org/10.1161/CIRCULATIONAHA.117.027597
Kitamura S (2011) Physiological and metabolic effects of grafts in coronary artery bypass surgery. Circ J 75(4): 766–772. https://doi.org/10.1253/circj.cj-10-1302
Carrel T, Winkler B (2017) Current trends in selection of conduits for coronary artery bypass grafting. Gen Thorac Cardiovasc Surg 65(10): 549–556. https://doi.org/10.1007/s11748-017-0807-8
Kalinin RE, Suchkov IA, Pshennikov AS, Vinogradov SA (2019) Markers of arteriovenous differentiation of endothelial cells and their influence on adaptation of autovenous conduits in main arteries reconstructive surgery. Surgery News 27(1): 91–100. https://doi.org/ 10.18484/2305-0047.2019.1.91
Malinska A, Podemska Z, Sujka-Kordowska P, Witkiewicz W, Nowicki M, Perek B, Witt M (2017) Caveolin 2: a facultative marker of unfavourable prognosis in long-term patency rate of internal thoracic artery grafts used in coronary artery bypass grafting. Preliminary report. Interact Cardiovasc Thorac Surg 24(5): 714–720. https://doi.org/10.1093/icvts/ivw411
Gaudino M, Di Franco A, Bhatt DL, Alexander JH, Abbate A, Azzalini L, Sandner S, Sharma G, Rao SV, Crea F, Fremes SE, Bangalore S (2021) The association between coronary graft patency and clinical status in patients with coronary artery disease. Eur Heart J 42(14): 1433–1441. https://doi.org/10.1093/eurheartj/ehab096
Spadaccio C, Antoniades C, Nenna A, Chung C, Will R, Chello M, Gaudino M (2020) Preventing treatment failures in coronary artery disease: what can we learn from the biology of in-stent restenosis, vein graft failure, and internal thoracic arteries? Cardiovasc Res 116(3): 505–519. https://doi.org/10.1093/cvr/cvz214
Gaudino M, Toesca A, Maggiano N, Pragliola C, Possati G (2003) Localization of nitric oxide synthase type III in the internal thoracic and radial arteries and the great saphenous vein: a comparative immunohistochemical study. J Thorac Cardiovasc Surg 125(6): 1510–1515. https://doi.org/10.1016/S0022-5223(03)00029-1
Broeders MA, Doevendans PA, Maessen JG, van Gorsel E, Egbrink MG, Daemen MJ, Tangelder GJ, Reneman RS, van der Zee R (2001) The human internal thoracic artery releases more nitric oxide in response to vascular endothelial growth factor than the human saphenous vein. J Thorac Cardiovasc Surg 122(2): 305–309. https://doi.org/10.1067/mtc.2001.113602
Tadjkarimi S, O’Neil GS, Luu TN, Allen SP, Schyns CJ, Chester AH, Yacoub MH (1992) Comparison of cyclic GMP in human internal mammary artery and saphenous vein: implications for coronary artery bypass graft patency. Cardiovasc Res 26(3): 297–300.
Ozen G, Aljesri K, Turkyilmaz G, Turkyilmaz S, Kavala AA, Topal G, Norel X (2022) Comparative study of coronary artery bypass graft materials: reduced contraction and ADMA levels in internal mammary artery versus saphenous vein. J Cardiovasc Surg (Torino) 63(1): 69–77. https://doi.org/10.23736/S0021-9509.21.11796-3
Funding
This study was supported by the Complex Program of Basic Research under the Siberian Branch of the Russian Academy of Sciences within the Basic Research Topic of Research Institute for Complex Issues of Cardiovascular Diseases № 0419-2021-001 «Novel anti-atherosclerotic therapies and machine learning solutions for automated diagnosis and prognostication of cardiovascular disease». The study is financially supported by the Ministry of Science and Higher Education of the Russian Federation (National Project Science and Universities).
Author information
Authors and Affiliations
Contributions
Idea of work and planning of the experiment—A.V.F., A.G.K., E.V.G., performing experiment—D.K.S., V.E.M., M.Yu.S., A.V.S., A.O.P., A.Yu.K., data processing—A.G.K., literature analysis—A.Yu.K., V.E.M., A.O.P., N.I.Z., writing and editing the article—A.V.F., V.E.M., A.Yu.K, A.G.K.
Corresponding author
Ethics declarations
CONFLICT OF INTEREST
The authors declare no apparent or potential conflicts of interest related to the publication of this article.
Additional information
Translated by A. Dyomina
Russian Text © The Author(s), 2022, published in Rossiiskii Fiziologicheskii Zhurnal imeni I.M. Sechenova, 2022, Vol. 108, No. 8, pp. 940–956https://doi.org/10.31857/S0869813922080039.
Rights and permissions
About this article
Cite this article
Frolov, A.V., Shishkova, D.K., Markova, V.E. et al. Paracrine Effects of Conditioned Medium during Its Cross-Addition to Arterial and Venous Endothelial Cells. J Evol Biochem Phys 58, 1180–1192 (2022). https://doi.org/10.1134/S0022093022040214
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022093022040214