Abstract
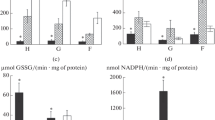
The state of the antioxidant (AO) system (AOS) and the content of products of lipid peroxidation (LPO) in hepatopancreas, gill, and foot of the Black Sea mollusc Mytilus galloprovincialis Lam. were studied in norm and under conditions of natural oxidative stress (spawning). It has been established that under conditions of spawning, the AO-protection of hepatopancreas cells is predominantly oriented to inactivation of high concentrations of peroxide compounds with participation of glutathione peroxidase and catalase. In the foot tissue, on the contrary, the main role in AO-processes is played by enzymatic inhibition of superoxide anion radical with aid of superoxide dismutase. The highest level of oxidative stress in the spawning state is revealed in the mollusc gill, which is indicated by the maximal increase of the content of TBA (thiobarbituric acid)-active products.
Similar content being viewed by others
References
Burdin, K.S., Krupina, M.V., and Saveliev, I.B., Molluscs of Mytilus Genus as Possible Indexes of the Content of Heavy and Transient Metals in Marine Water, Okeanologiya, 1979, vol. 19, pp. 1038–1044.
Muller, M., Pyocyonin Induces Oxidative Stress in Human Endothelial Cells and Modulates the Glutathione Redox Cycle, Free Rad. Biol. Med., 2002, vol. 33, pp. 1527–1533.
Dolmatova, L.S., Eliseikina, M.G., and Romashina, V.V., Antioxidant Enzyme Activity of Coelomocytes in the Far East Sea-Cucumber Eupentacta fraudarix, Zh. Evol. Biokhim. Fiziol., 2004, vol. 40, pp. 105–111.
Leus, Yu.V. and Grubinko, V.V., Activity of the Carp Antioxidant System during Action of Heavy Metal Ions, Gidrobiol. Zh., 1998, vol. 34, pp. 59–63.
Livingstone, D.R., Lemaire, Ph., Matthews, A., Peters, L.D., Porte, C., Fitzpatrick, P.J., Forlin, L., Nasci, C., Fossato, V., Wooton, N., and Goldfarb, P., Assessment of the Impact of Organic Pollutants on Goby (Zosterisessor ophiocephalus) and Mussel (Mytilus galloprovincialis) from the Venice Lagoon, Italy: Biochemical Studies, Mar. Environ. Res., 1995, no. 39, pp. 235–240.
Obrosova, I. G., Fathallah, L., Liu, E., and Nourooz-Zadeh, J., Early Oxidative Stress in the Diabetic Kidney: Effect of DL-α-Lipoic Acid, Free Rad. Biol. Med., 2003, vol. 34, pp. 186–195.
Gamble, S.C., Goldfarb, P.S., Porte, C., and Livingstone, D.R., Glutathione Peroxidase and Other Antioxidant Enzyme Function on Marine Invertebrates (Mytilus edulis, Pecten maximus, Garcinus maenas and Asterias rubens), Mar. Environ. Res., 1995, no. 39, pp. 191–195.
Yuneva, M.O., Characteristics of Antioxidant System in Mice of the SAM Strain (Senescence Accelerated Mice), Candidate Dissertation, Moscow, 2001.
Canesi, L., Viarengo, A., Leonzio, A., Filippelli, M., and Gallo, G., Heavy Metals and Glutathione Metabolism in Mussel Tissues, Aquat. Toxicol., 1999, vol. 46, pp. 67–76.
Romeo, M., Khessiba, A., Hoarau, P., Gnassia-Barelli, M., and Aissa, P., Biochemical Responses from Mussel Mytilus galloprovincialis of Bizerte (Tunisia) to Pollutants Exposition, J. Rech. Oceanogr., 2001, vol. 26, pp. 52–54.
Lionetto, M.G., Caricato, R., Giordano, M.E., Pascariello, M.F., Marinosci, L., and Schettino, I., Integrated Use of Biomarkers (Acetylcholinesterase and Antioxidant Enzymes Activities) in Mytilus galloprovincialis and Mullus barbatus in an Italian Coastal Marine Area, Mar. Pollut. Bull., 2003, vol. 46, pp. 324–330.
Isani, G., Serra, R., Cattani, O., et al., Adenylate Energy Charge and Metallothionein as Stress Indices in Mussels Exposed to Anoxia, J. Mar. Biol. Assoc. U.K., 1997, vol. 77, pp. 1187–1197.
Pellerin-Massicotte, Araujo M., Influence of Elevated Temperature and Air-Exposed in MDA Levels and Catalase Activities in Digestive Gland of the Blue Mussel (Mytilus edulis L.), J. Rech. Oceanogr., 1997, vol. 22, pp. 91–98.
Canesi, L. and Viarengo, A., Age-Related Differences in Glutathione Metabolism in Mussel Tissues (Mytilus edulis L.), Comp. Biochem. Physiol., 1997, vol. 116B, pp. 217–221.
Pirkova, A.V., Reproduction of Mussel Mytilus galloprovincialis Lam. and Some Aspects of Biotechnology of Its Cultivation, Candidate Dissertation, Sevastopol, 1994.
Pereslegina, I.A., Activities of Antioxidant Enzymes in Saliva of Healthy Children, Lab. Delo, 1989, no. 11, pp. 20–23.
Girin, S.V., Modification of Method of Determination of Catalase Activity in Biological Substrates, Lab. Diagnostika, 1999, no. 4, pp. 45–46.
Praktikum po immunologii. Uchebnoe posobie dlya studentov vuzov (Practical Work on Immunology: The Manual for High School Students), Frommel, A., Ed., Moscow, 1989.
Putilina, F.E., Determination of the Reduced Glutathione Content in Tissues, Metody biokhimicheskikh issledovanii (Methods in Biochemical Studies), Leningrad, 1982, pp. 183–186.
Ohkawa, И., Ohishi, N., and Yagi, K., Assay for Lipid Peroxides in Animal Tissues by Thiobarbituric Acid Reaction, Analyt. Biochem., 1979, vol. 95, pp. 351–358.
Lowry, O.H., Rosebrough, N.J., Farr, A.L., et al., Protein Measurement with the Folin Phenol Reagent, J. Biol. Chem., 1951, vol. 193, p. 75.
Menshikova, E.B. and Zenkov, N.K., Antioxidants and Inhibitors of Radical Oxidative Processes, Usp. Sovr. Biol., 1993, vol. 113, pp. 442–455.
White, A., Handler, P., Smith, E.L., Hill, R.L., and Lehman, I.R., Osnovy biokhimii (Fundamentals of Biochemistry), Moscow, 1981, vol. 1.
Bondar’, T.N., Pankin, V.Z., and Antonovskii, V.L., Reduction of Organic Hydroperoxide Compounds by Glutathione Peroxidase and Glutathione-S-Transferase: Effect of Substrate Structure, Dokl. AN SSSR, 1989, vol. 304, pp. 216–220.
Lukiyanova, L.D., Balamukhanov, B.S., and Ugolev, A.T., Kislorodzavisimye protsessy v kletke i ee funktsionalnoe sostoyanie (Oxygen-Dependent Processes in the Cell and Its Functional State), Moscow, 1982.
Dubinina, E.E. and Shugalei I.V., Oxidative Modification of Proteins, Usp. Sovr. Biol., 1993, vol. 113, no. 1, pp. 71–81.
Kulinskii, V.I. and Kolesnichenko, L.S., Structure, Properties, Biological Role and Regulation of Glutathione Peroxidase, Usp. Sovr. Biol., 1993, vol. 113, pp. 107–122.
Ivanov, V.N., Kholodov, V.I., Senicheva, M.I., et al., Biologiya kul’tiviruemykh midii (Biology of Cultivated Mussels), Kiev, 1989.
Lucas, J.M., Vaccaro, E., and Waite, J.H., A Molecular, Morphometric and Mechanical Comparison of the Structural Elements of Byssus from Mytilus edulis and Mytilus galloprovincialis, J. Exp. Biol., 2002, vol. 205, pp. 1807–1817.
Coyne, K.J. and Waite, J.H., In Search of Molecular Dovetails in Mussel Byssus: from the Threads to the Stem, J. Exp. Biol., 2000, vol. 203, pp. 1425–1431.
Carrington, E., Seasonal Variation in the Attachment Strength of Blue Mussels: Causes and Consequences, Limnol. Oceanogr., 2002, vol. 47, pp. 1723–1733.
Chelomin, V.P., Belcheva, N.N., and Zakhartsev, M.V., Biochemical Mechanisms of Adaptation of Mussel Mytilus trossulus to Cadmium and Copper Ions, Biol. Morya, 1998, vol. 24, pp. 319–325.
Lukiyanova, O.N., Molecular Biomarkers of Power Exchange in Mussels at Anthropogenic Pollution of Peter-the-Great Gulf of the Japanese Sea, Ekologiya, 2006, no. 3, pp. 227–231.
Chuiko, G.M., Causes of Interspecies Differences in Resistance of Bony Fish to Action of Organophosphorus Pesticides, Sovremennye problemy vodnoi toksikologii (Current Problems in Aquatic Toxicology: Abstracts of All-Russia Conference with Participation of Experts from the Near and Far Foreign Countries, November 19–21, 2002), Borok, 2002, p. 69.
Author information
Authors and Affiliations
Additional information
Original Russian Text © A. A. Soldatov, O. L. Gostyukhina, I. V. Golovina, 2008, published in Zhurnal Evolyutsionnoi Biokhimii i Fiziologii, 2008, Vol. 44, No. 2, pp. 150–155.
Rights and permissions
About this article
Cite this article
Soldatov, A.A., Gostyukhina, O.L. & Golovina, I.V. State of the antioxidant enzyme complex in tissues of the Black Sea mollusc Mytilus galloprovincialis under natural oxidative stress. J Evol Biochem Phys 44, 175–182 (2008). https://doi.org/10.1134/S0022093008020047
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022093008020047